Housekeeping: Happy New Year!! No, I haven't graded your quizzes because when I go on break, that's exactly what I do...BREAK! You'll get them whenever I finish grading, so please don't worry me about it.
We are now on Section 2.2. We should finish this section by next week and I will quiz you over it. The dates for quiz and exam are already on the testing calendar. As far as the next lab, I am looking for database work for you to complete, as dB work counts as lab hours.
Agenda:
1. Review of the atom
2. The electromagnetic spectrum
3. The Bohr Model
4. Atomic Orbitals (I)
Lesson Objectives:
1. Explain the phenomenon of emission spectra.
2. Describe the line emission spectrum of hydrogen and its relationship to the Bohr Model.
3. Describe the relationship between color, wavelength, frequency and energy across the eMag spectrum.
4. Calculate either frequency or wavelength using the speed of light equation.
Content Review:
Atomic Theory Structure Electron Behavior Electron Configuration
Student Missions:
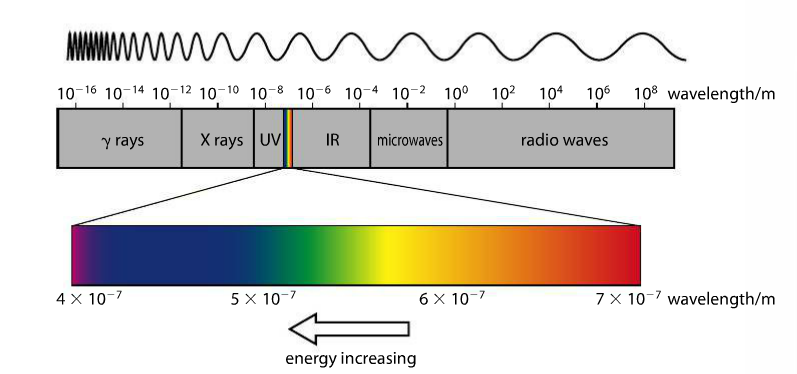
Mission 1: It's All About Colors! The electromagnetic spectrum is a visualization of electromagnetic radiation. It ranges from low energy radio waves to high energy gamma rays. All eMag waves travel at the same speed (c) but are distinguished by their different wavelengths (greek letter lambda). Different colors of visible light (the sliver of the spectrum we can see) have different wavelengths. There is a full eMag spectrum in your Data Booklet.
We are now on Section 2.2. We should finish this section by next week and I will quiz you over it. The dates for quiz and exam are already on the testing calendar. As far as the next lab, I am looking for database work for you to complete, as dB work counts as lab hours.
Agenda:
1. Review of the atom
2. The electromagnetic spectrum
3. The Bohr Model
4. Atomic Orbitals (I)
Lesson Objectives:
1. Explain the phenomenon of emission spectra.
2. Describe the line emission spectrum of hydrogen and its relationship to the Bohr Model.
3. Describe the relationship between color, wavelength, frequency and energy across the eMag spectrum.
4. Calculate either frequency or wavelength using the speed of light equation.
Content Review:
Atomic Theory Structure Electron Behavior Electron Configuration
Student Missions:
Mission 1: It's All About Colors! The electromagnetic spectrum is a visualization of electromagnetic radiation. It ranges from low energy radio waves to high energy gamma rays. All eMag waves travel at the same speed (c) but are distinguished by their different wavelengths (greek letter lambda). Different colors of visible light (the sliver of the spectrum we can see) have different wavelengths. There is a full eMag spectrum in your Data Booklet.
This image from the Pearson text (p. 71) shows the changing wavelength of the eMag spectrum in meters (m). On the left side, we have high energy gamma waves, which have the shortest wavelengths, and on the right side, we have radio waves, which have the longest wavelengths. The sliver of color between UV and IR is what our eye sees, magnified as the bar of color directly above.
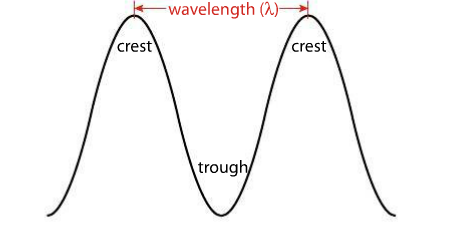
Wavelength is measured from the crest (or trough) of one wave to the crest (or trough) of another. The number of waves that pass a given point at any time is called frequency. The relationship between frequency and wavelength is inverse: the shorter the wavelength, the higher the frequency.
Wavelength is measured from the crest (or trough) of one wave to the crest (or trough) of another. The number of waves that pass a given point at any time is called frequency. The relationship between frequency and wavelength is inverse: the shorter the wavelength, the higher the frequency.
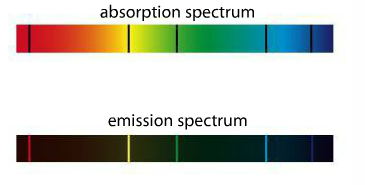
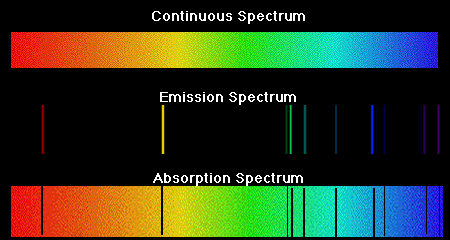
| When eMag radiation is passed through atoms, some of the radiation is absorbed and used to excite the atoms from low energy levels to high energy levels. The mass spectrometer analyses the transmitted radiation relative to the incident radiation and an absorption spectrum is produced. Absorption spectra show colors with black lines and the black lines represent where the energy is absorbed. Emission spectra result when a high voltage is applied to the gas. Emission spectra show black with a few colored lines. The colored lines are the same as those that are missing from the absorption spectrum. They read like bar codes to identify unknown elements. |
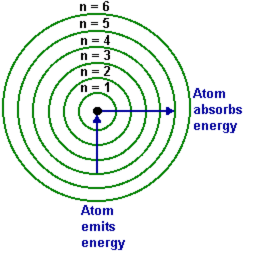
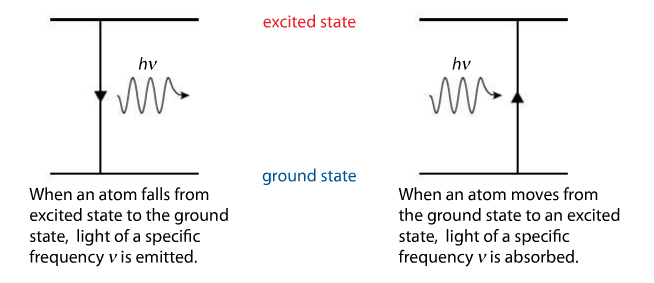
| Niels Bohr proposed that when an atom absorbs energy, the electron (we are specifically referencing hydrogen) moves to a higher energy level. This is called the excited state, which is unstable. Soon, the electron returns to the lowest level, aka the ground state. The energy the electron gives off when it returns to the ground state is released in the form of eMag energy. A packet of energy, called a photon, is released for each electron transition. To the left, you see a Bohr model with numbered energy levels. Photons that release UV light will have more energy than photons of IR light. The energy is proportional to the frequency of the radiation. The lower image can be found on p. 72. The energy of the photon is equal to the energy change in the atom AND the frequency of the radiation. |
Homework: Page 75, #12-14. I screenshot'd the page and stuck it below, so no excuses. I also want you to attempt the challenge question.
Mission 2: SUPERMODELS! The Bohr model is the standard, and even though it is outdated, it still provides a simplistic understanding of how electrons behave when energized. Research done by Werner Heisenberg & Erwin Schrodinger in the 1920s & 1930s produced a different understanding of the atom.
The Bohr model fails with elements beyond hydrogen because it assumes that the electron's trajectory can be precisely described. This is not true because any attempt to measure an electron's position disturbs its motion or velocity. This principle is called Heisenberg's Uncertainty Principle: an electron's position and velocity cannot be known simultaneously because manipulating one affects the other. The best that can be hoped for is a probability of where the electron is likely to be.
Schrodinger came along and, using the principles of wave/particle theory, proposed that a wave equation can be used to describe electron behavior in the same way light behavior is described. The equation works for hydrogen and elements beyond hydrogen, and the solutions to the equation are known as atomic orbitals. These are regions of space around the nucleus in which there's a 90% chance of finding an electron. Orbital shape depends on the energy of the electron.
The Bohr model fails with elements beyond hydrogen because it assumes that the electron's trajectory can be precisely described. This is not true because any attempt to measure an electron's position disturbs its motion or velocity. This principle is called Heisenberg's Uncertainty Principle: an electron's position and velocity cannot be known simultaneously because manipulating one affects the other. The best that can be hoped for is a probability of where the electron is likely to be.
Schrodinger came along and, using the principles of wave/particle theory, proposed that a wave equation can be used to describe electron behavior in the same way light behavior is described. The equation works for hydrogen and elements beyond hydrogen, and the solutions to the equation are known as atomic orbitals. These are regions of space around the nucleus in which there's a 90% chance of finding an electron. Orbital shape depends on the energy of the electron.
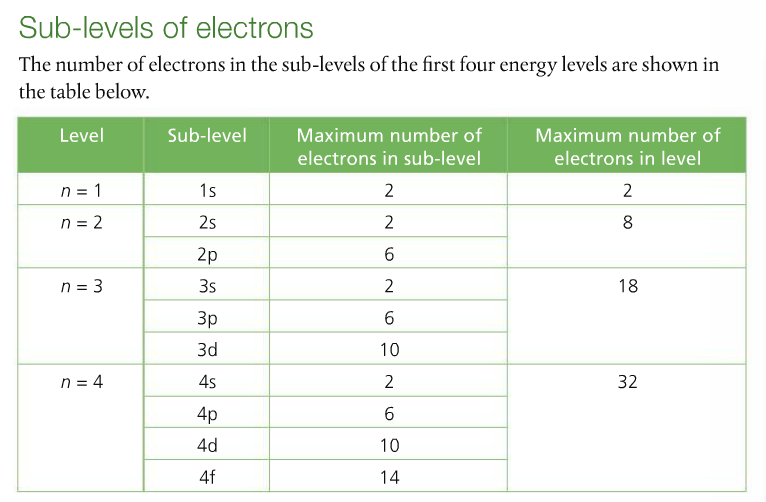
Make note of the chart that she draws to denote the relationship between principal energy level, sublevel, & number of electrons held. This information corresponds to pages 76-77 and is much easier to understand. You are expected to know the shapes and names of s and p atomic orbitals.
Mission 3: There Are Always Rules...There are three rules for writing electron configurations and drawing orbital diagrams. They are listed here. Take a look and make sure you understand each one.
This is the shorthand version of all this information: When describing the arrangement of an element's electrons, it is important to understand that (1) you must begin at the ground state, which is the energy level closest to the nucleus (n = 1) and work your way up, (2) there are four sublevels (s p d f) within each principal energy level; each sublevel has a particular shape called an orbital, and (3) each orbital can hold a maximum of two electrons spinning in opposite directions.
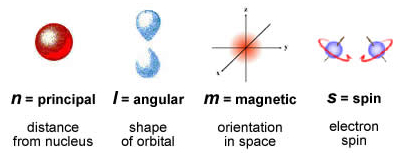
This model of the atom, compiled from the research of Schrodinger, Heisenberg, and a few others, is called the Quantum Mechanical Model. It is a mathematical model explained by four quantum numbers.
Image courtesy of crescentok.com.
Mission 3: There Are Always Rules...There are three rules for writing electron configurations and drawing orbital diagrams. They are listed here. Take a look and make sure you understand each one.
This is the shorthand version of all this information: When describing the arrangement of an element's electrons, it is important to understand that (1) you must begin at the ground state, which is the energy level closest to the nucleus (n = 1) and work your way up, (2) there are four sublevels (s p d f) within each principal energy level; each sublevel has a particular shape called an orbital, and (3) each orbital can hold a maximum of two electrons spinning in opposite directions.
This model of the atom, compiled from the research of Schrodinger, Heisenberg, and a few others, is called the Quantum Mechanical Model. It is a mathematical model explained by four quantum numbers.
Image courtesy of crescentok.com.
Mission 4: Orbital Diagrams & Electron Configuration. I'm just going to drop this right here...
Let's Practice!! You need to know the electron configurations/orbital diagrams of elements 1 - 36, so let's get it!



 RSS Feed
RSS Feed