C2: Fossil Fuels
The formation of fossil fuels from decaying organisms is an example of reduction. Recall that reduction is the gain of hydrogen with the loss of oxygen. Many fossil fuels contain saturated alkanes and during the formation of fossil fuels, carbon atoms became more and more saturated with hydrogen and have fewer bonds to nitrogen, sulfur and/or oxygen than existed in the living form.
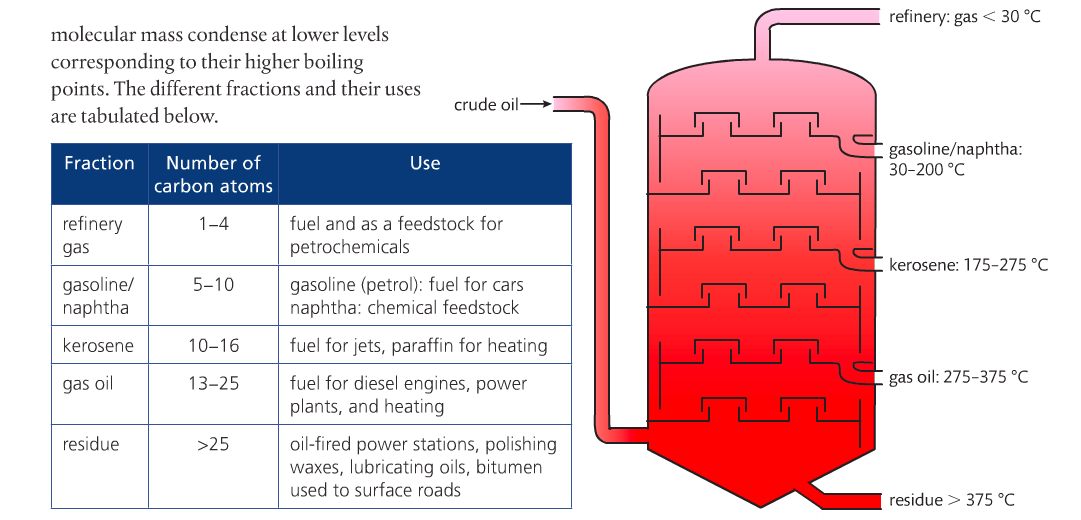
There are three main fossil fuels: coal, gas and crude oil. Coal is the most abundant but crude oil, also known as petroleum, is the most important. However, petroleum is difficult to use in its natural form. Petroleum contains a mixture of hydrocarbons of varying lengths. Long chain hydrocarbons are stronger intermolecular forces than short chain hydrocarbons. As a result, boiling points can be used to separate crude oil into fractions of varying chain lengths. At oil refineries, the fractions are separated by fractional distillation.
First image courtesy of the Pearson text. Second image comes from the Oxford text.
There are three main fossil fuels: coal, gas and crude oil. Coal is the most abundant but crude oil, also known as petroleum, is the most important. However, petroleum is difficult to use in its natural form. Petroleum contains a mixture of hydrocarbons of varying lengths. Long chain hydrocarbons are stronger intermolecular forces than short chain hydrocarbons. As a result, boiling points can be used to separate crude oil into fractions of varying chain lengths. At oil refineries, the fractions are separated by fractional distillation.
First image courtesy of the Pearson text. Second image comes from the Oxford text.
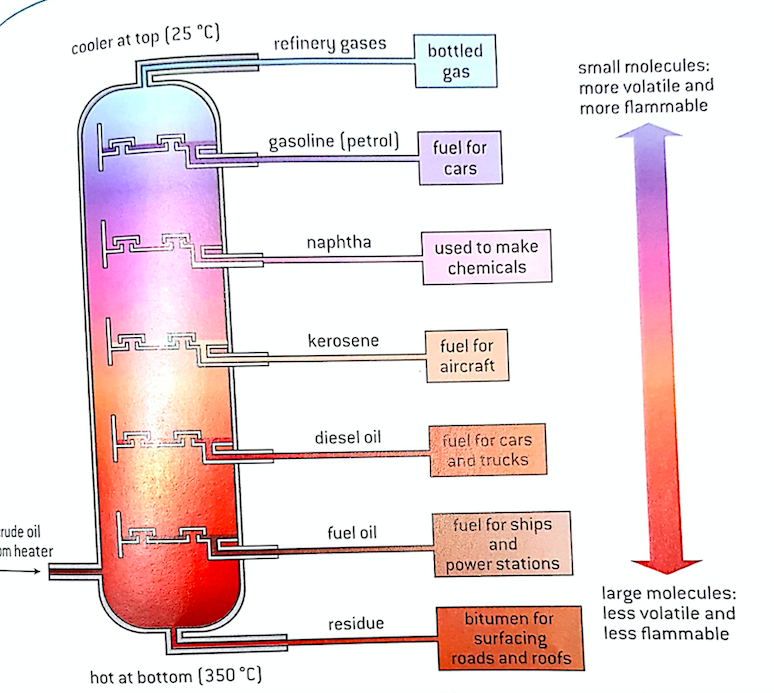
The crude oil is heated and becomes less viscous. Temperatures are lower at the top, so substances with low boiling points leave the column whereas fractions with higher BPs condense at higher temperatures near the bottom. The longer chain hydrocarbons are more viscous, darker in color and less volatile, therefore they are less flammable than the shorter chains hydrocarbons.
The more volatile short chain hydrocarbons make better fuels and burn cleaner. However, there are more long chain hydrocarbons in crude oil than short ones. In order to get more short chains, a process called cracking is used. This process is extremely important in the production of gasoline and diesel fuel.
The more volatile short chain hydrocarbons make better fuels and burn cleaner. However, there are more long chain hydrocarbons in crude oil than short ones. In order to get more short chains, a process called cracking is used. This process is extremely important in the production of gasoline and diesel fuel.
When fuels are burned in automobile engines, they are compressed and then lit with a spark. Some hydrocarbons have a higher tendency to auto-ignite, which produces an effect called knocking. This can damage an engine. A measure of the fuel's ability to resist auto-ignition is its octane rating. Higher octane fuels can be compressed more and give a better performance than fuels with lower octane ratings. Read pages 660-661 to find out more about the relationship between octane rating and and branched-chain hydrocarbons.
Catalytic reforming is used to convert low-octane numbered alkanes such as heptane or octane into higher-octane numbered isomers such as methylbenzene or 2,2,4-trimethylpentane. I strongly suggest watching Mr. Thornley's YouTube videos on cracking and reforming petroleum.
Catalytic reforming is used to convert low-octane numbered alkanes such as heptane or octane into higher-octane numbered isomers such as methylbenzene or 2,2,4-trimethylpentane. I strongly suggest watching Mr. Thornley's YouTube videos on cracking and reforming petroleum.
Coal is the most abundant fossil fuel. It is formed from the remains of plant life that have been subjected to geological heat and pressure. It can be converted to more useful forms that are cheaper than crude oil. One method is coal gasification in which synthesis gas (aka coal gas or syngas) is produced by reacting coal with oxygen and steam in a gasifier to create hydrocarbons. Coal gasification can occur in an underground cavity, giving low plant costs as no gasifier needs to be constructed, no coal needs to be brought to the surface, and the carbon dioxide formed can be stored underground. This process is an example of coal capture and storage and reduces the amount of carbon dioxide entering the atmosphere. Examine Figure 8 and Table 2 on page 663 in your text.
Coal liquefaction takes filtered and cleaned synthesis gas and adds water or carbon dioxide over a catalyst. This is known as indirect coal liquefaction (ICL). In direct coal liquefaction (DCL), hydrogen is added to heated coal in the presence of a catalyst. Both methods adjust the carbon-to-hydrogen ratio and produce synthetic liquid fuels known as the Fischer-Tropsch process. The equation is on page 663 in your text.
The production of energy by burning fuels produces carbon dioxide. The carbon foot print of a reaction is a measure of the net quantity of carbon dioxide produced by the process. Even though biofuels may cost more to produce, their carbon footprint is less because carbon dioxide is absorbed by photosynthesis while the fuel is growing.
Be sure to examine the worked problems in the textbook.
The production of energy by burning fuels produces carbon dioxide. The carbon foot print of a reaction is a measure of the net quantity of carbon dioxide produced by the process. Even though biofuels may cost more to produce, their carbon footprint is less because carbon dioxide is absorbed by photosynthesis while the fuel is growing.
Be sure to examine the worked problems in the textbook.


 RSS Feed
RSS Feed