Housekeeping: We will finish acids & bases this week. You have a relatively short quiz over the first three sections. Your exam is scheduled for next Monday.
Content Review:
Textbook: Chapter 8
Links: IB Chemistry Home Notes
Agenda:
1. Strong and weak acids and bases
2. Acid deposition
3. Practice problems/review
Mission 4: What Are You, Strong or Weak?
Mission Objectives. You should be able to...
1. Differentiate between strong and weak acids and bases.
2. List the properties of strong and weak acids and bases.
Strong acids and bases disassociate completely in water. This means that the acid/base breaks up completely into their component ions. By contrast, weak acids and bases disassociate only partially. If you look at the ionization of a strong acid/base, you will see that it is a forward reaction only. Examining the ionization of a weak acid/base requires an equilibrium reaction.
Properties of strong acids & bases:
1. Strong acids/bases conduct electricity very well due to the number of ions in solution. 2. Reaction rates using strong acids/bases increase due to the number of ions in solution.
3. For strong acids, the higher the [H+], the lower the pH. For strong bases, the lower the [H+], the higher the pH.
4. Strong acids are good proton donors and have weak conjugate bases.
5. Strong bases are good proton acceptors and have weak conjugate acids.
Strong acids: hydrochloric acid, nitric acid, sulfuric acid. All others are WEAK.
Strong bases: sodium hydroxide, potassium hydroxide, rubidium hydroxide and barium hydroxide. All others are WEAK.
Content Review:
Textbook: Chapter 8
Links: IB Chemistry Home Notes
Agenda:
1. Strong and weak acids and bases
2. Acid deposition
3. Practice problems/review
Mission 4: What Are You, Strong or Weak?
Mission Objectives. You should be able to...
1. Differentiate between strong and weak acids and bases.
2. List the properties of strong and weak acids and bases.
Strong acids and bases disassociate completely in water. This means that the acid/base breaks up completely into their component ions. By contrast, weak acids and bases disassociate only partially. If you look at the ionization of a strong acid/base, you will see that it is a forward reaction only. Examining the ionization of a weak acid/base requires an equilibrium reaction.
Properties of strong acids & bases:
1. Strong acids/bases conduct electricity very well due to the number of ions in solution. 2. Reaction rates using strong acids/bases increase due to the number of ions in solution.
3. For strong acids, the higher the [H+], the lower the pH. For strong bases, the lower the [H+], the higher the pH.
4. Strong acids are good proton donors and have weak conjugate bases.
5. Strong bases are good proton acceptors and have weak conjugate acids.
Strong acids: hydrochloric acid, nitric acid, sulfuric acid. All others are WEAK.
Strong bases: sodium hydroxide, potassium hydroxide, rubidium hydroxide and barium hydroxide. All others are WEAK.
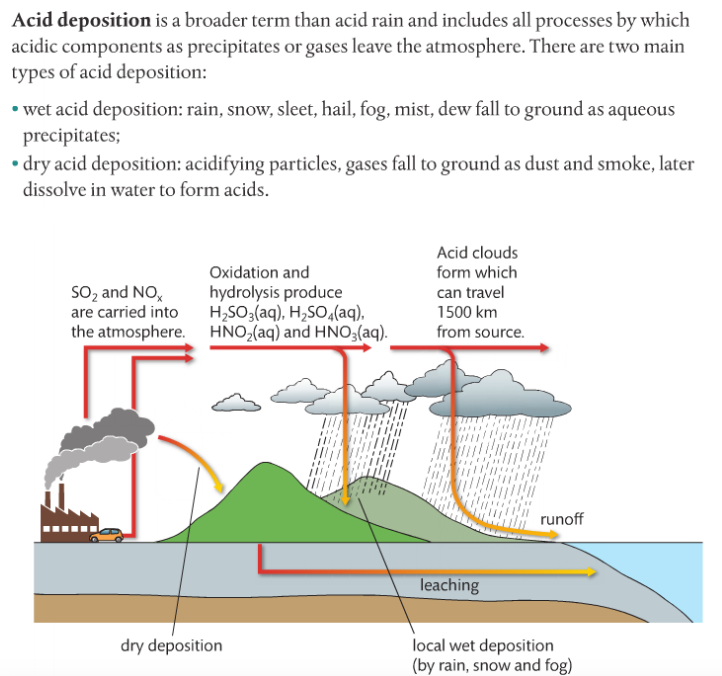
Mission 2: Acid Deposition.
Mission Objectives. You should be able to...
1. List the compounds that make up acid rain.
2. Describe the sources of the oxides of sulfur and nitrogen.
3. State the effects of acid deposition.
Mission Objectives. You should be able to...
1. List the compounds that make up acid rain.
2. Describe the sources of the oxides of sulfur and nitrogen.
3. State the effects of acid deposition.
We will spend the rest of the period working problems and reviewing for the quiz and exam. Remember the quiz is Thursday and the exam is the following Monday.




 RSS Feed
RSS Feed