Housekeeping: We will be titrating this week. Today, after you finish last Friday's lab, I will introduce the topic. We will practice titrating during Thursday's class, and on Friday, you will do it for real. We should finish the acid/base unit by the end of next week.
Content Review:
Textbook: Chapter 8
Links: IB Chemistry Home Notes
Agenda:
1. Acid-base titration
2. The pH scale
Mission 1: Acid-Base Titration
Mission Objectives. You should be able to:
1. Understand the nature of titration.
2. Titrate a known acid with an unknown base.
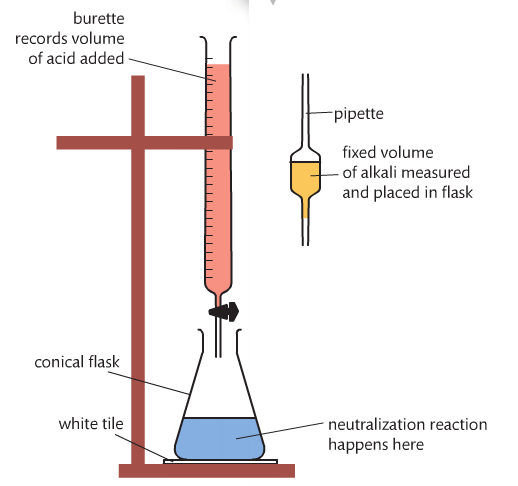
A titration is a volumetric analysis technique that involves a reaction between a solution of unknown concentration (c) and a standardized solution (titrant). The titrant is in the buret and the unknown concentration is in the flask. The objective is to determine the unknown concentration by reaching the equivalence point (when the acid neutralizes the base).
Content Review:
Textbook: Chapter 8
Links: IB Chemistry Home Notes
Agenda:
1. Acid-base titration
2. The pH scale
Mission 1: Acid-Base Titration
Mission Objectives. You should be able to:
1. Understand the nature of titration.
2. Titrate a known acid with an unknown base.
A titration is a volumetric analysis technique that involves a reaction between a solution of unknown concentration (c) and a standardized solution (titrant). The titrant is in the buret and the unknown concentration is in the flask. The objective is to determine the unknown concentration by reaching the equivalence point (when the acid neutralizes the base).
Here is a writeup of a titration lab. Review the materials list and the procedure before Thursday.
The first video goes through the setup of a titration. The second video shows an actual titration and the math that goes along with it.
The first video goes through the setup of a titration. The second video shows an actual titration and the math that goes along with it.
Mission 2: All About That pH!
Mission Objectives. You should be able to:
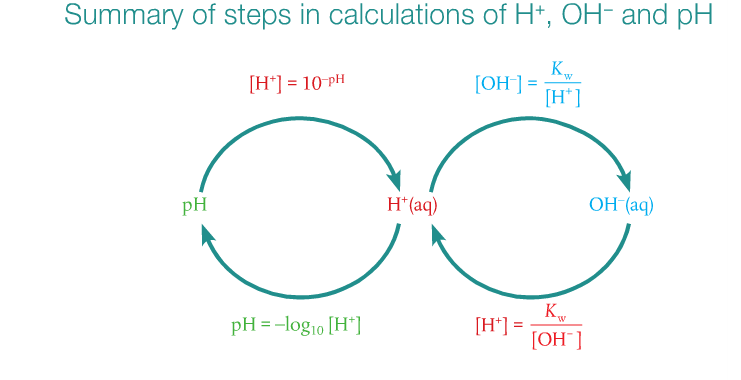
1. Calculate pH and pOH given the formula.
2. Understand that a change of one on a pH scale represents a change of 10X in the [H+].
3. Explain the arrangement of the pH scale in terms of acidity, neutrality and alkalinity.
We will start this lesson with an overview. Mr. Thornley tagged Mr. Anderson and so now he will jump into the ring and do his thing. We will work on some practice problems in class and for homework. It's important that you be able to calculate pH and pOH.
Mission Objectives. You should be able to:
1. Calculate pH and pOH given the formula.
2. Understand that a change of one on a pH scale represents a change of 10X in the [H+].
3. Explain the arrangement of the pH scale in terms of acidity, neutrality and alkalinity.
We will start this lesson with an overview. Mr. Thornley tagged Mr. Anderson and so now he will jump into the ring and do his thing. We will work on some practice problems in class and for homework. It's important that you be able to calculate pH and pOH.
 RSS Feed
RSS Feed