Housekeeping: Happy New Year, guys! We will be studying Chapter 5: Energetics & Thermochemistry this month.
Agenda:
1. Heat vs Temperature
2. Enthalpy
3. Specific Heat
4. Hess' Law
5. Bond Enthalpy
Content Review:
Text: Chapter 5
Links: The Physics Classroom The Nature of Energy Measuring Energy Changes
Student Missions:
Mission 1: Energy. Some Days Are Better Than Others.
Mission Objectives. You should be able to:
1. Explain how heat is a form of energy
2. Describe temperature as a measurement of kinetic energy
3. Explain the conservation of energy in chemical reactions
4. Compare endothermic and exothermic reactions
5. Explain the concept of system and surroundings
Some things you should know. Temperature and heat are not the same thing. Heat is energy that results from a temperature change and produces an increase in disorder in how particles behave. Heat increases the average kinetic energy of molecules, which is measured by temperature. Professor Dave, aka Chemistry Jesus, explains it all.
Agenda:
1. Heat vs Temperature
2. Enthalpy
3. Specific Heat
4. Hess' Law
5. Bond Enthalpy
Content Review:
Text: Chapter 5
Links: The Physics Classroom The Nature of Energy Measuring Energy Changes
Student Missions:
Mission 1: Energy. Some Days Are Better Than Others.
Mission Objectives. You should be able to:
1. Explain how heat is a form of energy
2. Describe temperature as a measurement of kinetic energy
3. Explain the conservation of energy in chemical reactions
4. Compare endothermic and exothermic reactions
5. Explain the concept of system and surroundings
Some things you should know. Temperature and heat are not the same thing. Heat is energy that results from a temperature change and produces an increase in disorder in how particles behave. Heat increases the average kinetic energy of molecules, which is measured by temperature. Professor Dave, aka Chemistry Jesus, explains it all.
Chemical reactions are accompanied by energy changes. Bonds are broken and formed in reactions, which correspond to an increase or decrease in energy, Energy is the ability to do work or move an object against an opposing force. Examples of energy are light, electricity, sound, heat, and chemical energy are absorbed or released during chemical reactions.
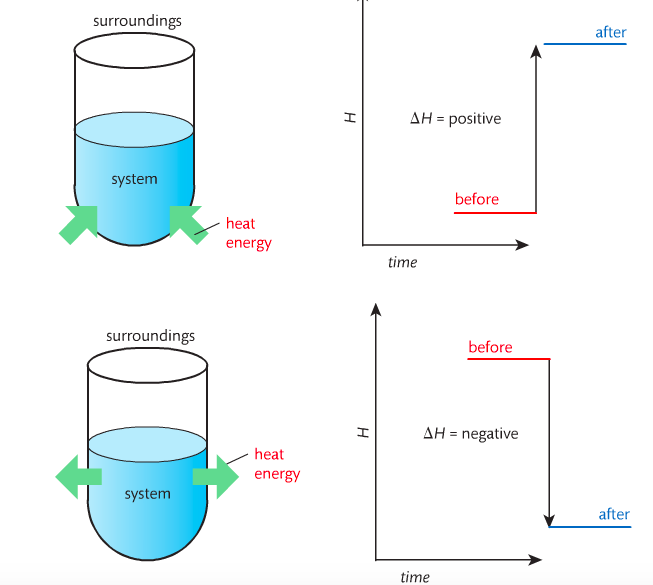
In order to do thermodynamic work, you need to understand the concept of system & surroundings. The system and surroundings must be defined in any example. The system is the area of interest and the surroundings include everything else. Most systems are open and interface with the surroundings. Some systems are closed and do not interface. The total energy in a system and its surroundings do not change.
In order to do thermodynamic work, you need to understand the concept of system & surroundings. The system and surroundings must be defined in any example. The system is the area of interest and the surroundings include everything else. Most systems are open and interface with the surroundings. Some systems are closed and do not interface. The total energy in a system and its surroundings do not change.
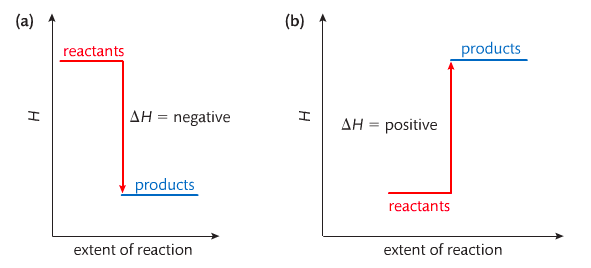
Heat contents of a system is called enthalpy A system is a heat reserve. Changes in energy are denoted by "delta H." Delta H is positive when heat is added and negative when heat is released.
Examine the diagrams on page 141. It shows the relationship between a system and its surroundings and how they look on an energy curve. Energy curves are endothermic or exothermic.
Enthalpy is stored in chemical bonds and intermolecular forces as potential energy. When substances react, the difference in the enthalpy between reactions and products produces an enthalpy change that can be observed.
Standard enthalpy changes are a set of conditions (which should be in your data booklet) are the following: 100 kPa, a concentration of 1 mol/dm3 (1M) for all solutions, and all substances in their standard states. Temperature is usually not a part of these conditions, but is normally included as 298K.
Most chemical reactions are exothermic; they give off heat and result in a transfer of energy from system to surroundings. Delta H is negative in exothermic reactions. Endothermic reactions absorb heat and result in a transfer of energy from surroundings to system. Delta H is positive in endothermic reactions.
Enthalpy is stored in chemical bonds and intermolecular forces as potential energy. When substances react, the difference in the enthalpy between reactions and products produces an enthalpy change that can be observed.
Standard enthalpy changes are a set of conditions (which should be in your data booklet) are the following: 100 kPa, a concentration of 1 mol/dm3 (1M) for all solutions, and all substances in their standard states. Temperature is usually not a part of these conditions, but is normally included as 298K.
Most chemical reactions are exothermic; they give off heat and result in a transfer of energy from system to surroundings. Delta H is negative in exothermic reactions. Endothermic reactions absorb heat and result in a transfer of energy from surroundings to system. Delta H is positive in endothermic reactions.
Mission 2: Let's Be Specific About This.
Mission Objectives. You should be able to:
1. Describe enthalpy changes and how they are expressed under standard conditions.
2. Calculate specific heat and enthalpy problems.
3. Write and interpret thermochemical equations.
A thermochemical equation is a balanced chemical equation that includes the enthalpy change (delta H). Remember that delta H is negative for exothermic reactions and positive for endothermic reactions.
Specific heat is the property of a substance which gives heat needed to increase the temperature by 1K. It depends on the number of particles present.
The formula for specific heat is
Mission Objectives. You should be able to:
1. Describe enthalpy changes and how they are expressed under standard conditions.
2. Calculate specific heat and enthalpy problems.
3. Write and interpret thermochemical equations.
A thermochemical equation is a balanced chemical equation that includes the enthalpy change (delta H). Remember that delta H is negative for exothermic reactions and positive for endothermic reactions.
Specific heat is the property of a substance which gives heat needed to increase the temperature by 1K. It depends on the number of particles present.
The formula for specific heat is
Practice Problems: One Two
Enthalpy Changes. The direction of change is in the direction of lower stored energy. Chemicals change in a way that reduces their enthalpy. It is expected that a reaction will occur if it leads to a reduction in enthalpy. Products in an exothermic reaction are more stable than the reactants, but stability is a relative term. The sign of delta H is a guide for the likely direction of change, but it is not 100% reliable. Endothermic reactions do occur when there is an increase in the disorder of a system; for example, when gases are produced.
Enthalpy Changes. The direction of change is in the direction of lower stored energy. Chemicals change in a way that reduces their enthalpy. It is expected that a reaction will occur if it leads to a reduction in enthalpy. Products in an exothermic reaction are more stable than the reactants, but stability is a relative term. The sign of delta H is a guide for the likely direction of change, but it is not 100% reliable. Endothermic reactions do occur when there is an increase in the disorder of a system; for example, when gases are produced.
Practice problems for Missions 1 & 2.
Mission 3: Hess' Law
Mission Objectives. You should be able to...
1. Complete enthalpy calculations using Hess' Law.
Hess' Law is an application of the conservation of energy law. A chemical equation shows the net reaction; it is a summary of a number of different reactions which are added together and result in an overall reaction. We will spend time working on questions in class.
Mission Objectives. You should be able to...
1. Complete enthalpy calculations using Hess' Law.
Hess' Law is an application of the conservation of energy law. A chemical equation shows the net reaction; it is a summary of a number of different reactions which are added together and result in an overall reaction. We will spend time working on questions in class.
Mission 4: Bond Enthalpy.
Mission Objectives. You should be able to...
1. Calculate the enthalpy changes from known bond enthalpy and comparison of these with experimentally measured values.
2. Sketch and evaluate potential energy profiles in determining whether reactants or products are more stable and if the reaction is endothermic or exothermic.
3. Discuss the bond strength in ozone relative to oxygen and its importance to the atmosphere.
Mr. Thornley is on the case. You will want to take notes from this video. He lays it out all clear and proper-like.
Mission Objectives. You should be able to...
1. Calculate the enthalpy changes from known bond enthalpy and comparison of these with experimentally measured values.
2. Sketch and evaluate potential energy profiles in determining whether reactants or products are more stable and if the reaction is endothermic or exothermic.
3. Discuss the bond strength in ozone relative to oxygen and its importance to the atmosphere.
Mr. Thornley is on the case. You will want to take notes from this video. He lays it out all clear and proper-like.
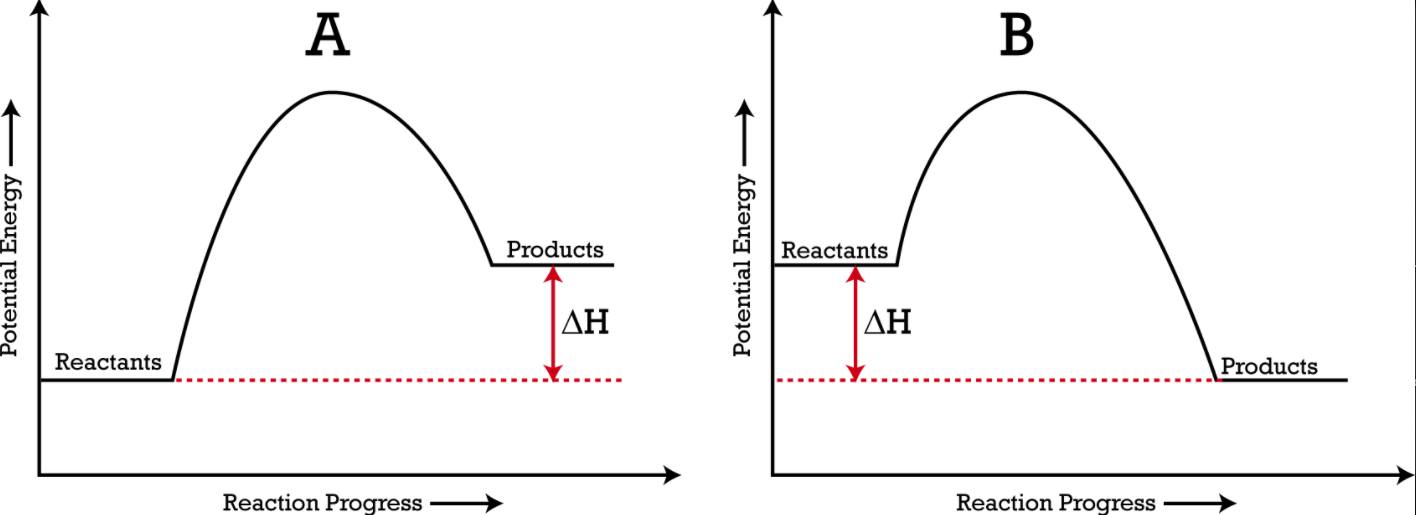
Below are two different energy profiles. The video that follows goes into detail about each profile. You need to make sure you are able to read and interpret each profile.
HOMEWORK: Practice problems from the text and the packet.


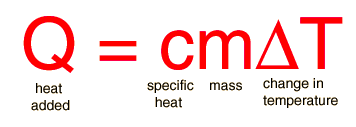


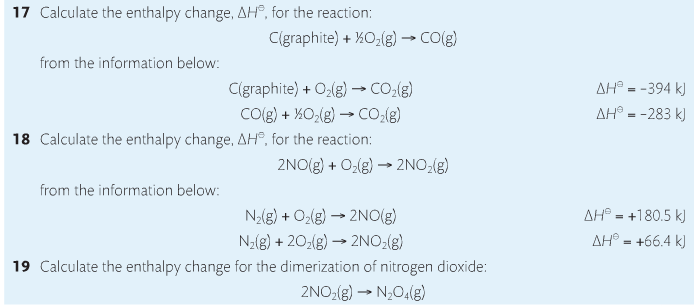
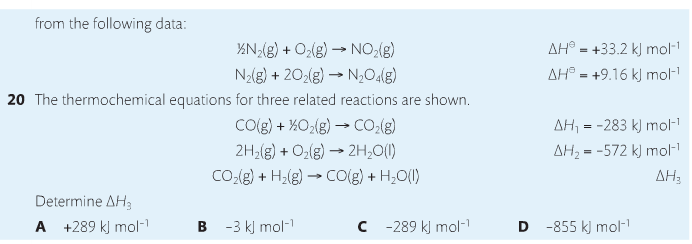
 RSS Feed
RSS Feed