Mission 1: First Row d-Block Elements
Mission Objectives. You should be able to...
1. Explain the ability of transition metals to form variable oxidation states from successive ionization energies.
2. Explain the nature of the coordinate bond within a complex ion.
3. Deduce the total charge given the formula of the ion and the ligands present.
4. Explain the magnetic properties in transition metals in terms of unpaired electrons.
Mission Objectives. You should be able to...
1. Explain the ability of transition metals to form variable oxidation states from successive ionization energies.
2. Explain the nature of the coordinate bond within a complex ion.
3. Deduce the total charge given the formula of the ion and the ligands present.
4. Explain the magnetic properties in transition metals in terms of unpaired electrons.
The IUPAC defines a transition metal as an element that has an incomplete d-sublevel, or can give rise to cations with incomplete d-sublevels. Based on this definition, group 12 elements (Zn, Cd, Hg, Cn) are not considered to be transition metals because they have full d-sublevels. This means that groups 3-11 are transition metals.
Characteristics of transition metals:
--variable oxidation states
--compounds and ions are colored
--form complexes with ligands
--often used as catalysts
--magnetic properties depend on oxidation states and coordination numbers
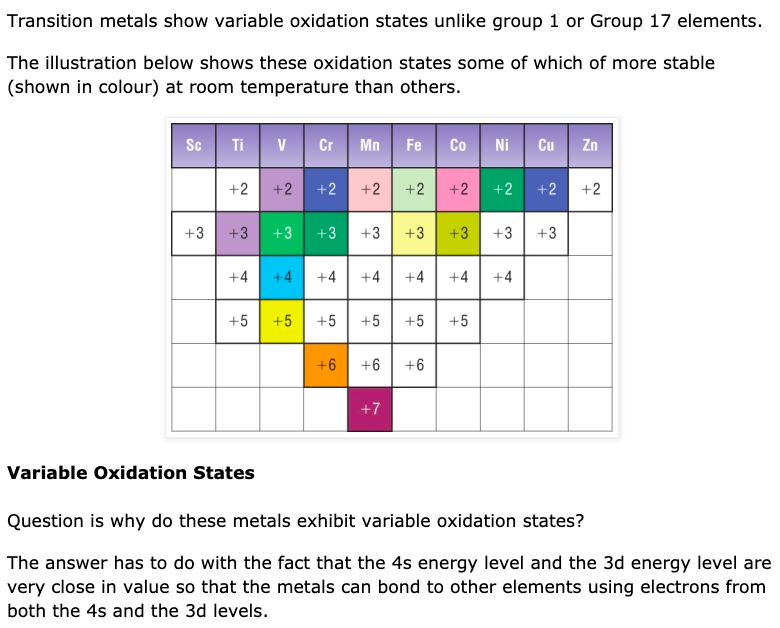
Look at elements manganese and chromium. Manganese can have oxidation states from +1 to +7. Mn+2 is colorless while Mn+7 is purple. See the below image from thesavvychemist.com.
Characteristics of transition metals:
--variable oxidation states
--compounds and ions are colored
--form complexes with ligands
--often used as catalysts
--magnetic properties depend on oxidation states and coordination numbers
Look at elements manganese and chromium. Manganese can have oxidation states from +1 to +7. Mn+2 is colorless while Mn+7 is purple. See the below image from thesavvychemist.com.
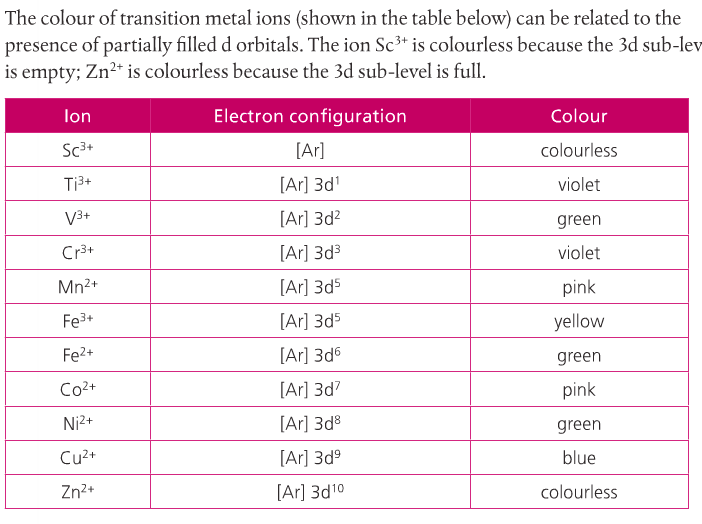
Your textbook lists some common colored compounds. Zinc compounds are usually colorless (due to the completely filled d-sublevel) unless the complex has a chromophore. The chromophore is a group of atoms responsible for the absorption of electromagnetic radiation, which can be absorbed in the visible region of the e-mag spectrum.
Transition metal complexes have a central metal atom bonded to a group of molecules or ions. These are called ligands. Ligands are molecules or ions with a lone pair of electrons that can attach themselves to ions with a positive charge density. Examples of ligands include water (H2O), ammonia (NH3), the cyanide ion (CN-), the hydroxide ion, (OH-), the oxalate ion (C2O42-), and EDTA. All of these molecules and ions have lone electron pairs.
Metal ions with a high charge density: Sn4+, Al3+, Cr3+, Cu2+, Zn2+, Be2+, Sn2+
Compounds are described as coordination compounds to signify the coordinate bonding present between ligands and the central metal ion. Monodentate ligands use one lone pair of electrons that form CCBs (coordinate covalent bond; also called dative covalent bond) with a central metal ion. Polydentate ligands have more than one lone pair of electrons to form CCBs to a central metal ion.
Coordination numbers are the total number of points at which a central atom or ion attaches ligands.
Transition metal complexes have a central metal atom bonded to a group of molecules or ions. These are called ligands. Ligands are molecules or ions with a lone pair of electrons that can attach themselves to ions with a positive charge density. Examples of ligands include water (H2O), ammonia (NH3), the cyanide ion (CN-), the hydroxide ion, (OH-), the oxalate ion (C2O42-), and EDTA. All of these molecules and ions have lone electron pairs.
Metal ions with a high charge density: Sn4+, Al3+, Cr3+, Cu2+, Zn2+, Be2+, Sn2+
Compounds are described as coordination compounds to signify the coordinate bonding present between ligands and the central metal ion. Monodentate ligands use one lone pair of electrons that form CCBs (coordinate covalent bond; also called dative covalent bond) with a central metal ion. Polydentate ligands have more than one lone pair of electrons to form CCBs to a central metal ion.
Coordination numbers are the total number of points at which a central atom or ion attaches ligands.
Read up on EDTA on p. 315. EDTA (EDTA4-) is a polydentate ligand that can form 6 CCBs to the central metal ion. It has a coordination number of 6. EDTA can remove metal ions from solution and inhibit enzyme-catalyzed reactions.
Transition metals are often used as catalysts in chemical reactions. Examples: the Haber Process, the decomposition of hydrogen peroxide, hydrogenation of alkenes, and hydrogenation of oils. They can be used in catalytic converters in cars and are biological catalysts. An example is heme, which is the iron center of hemoglobin. Hemoglobin is the protein that transports oxygen in the blood. Blood is red due to the presence of heme. Each subunit of hemoglobin has an iron atom at its center to which oxygen binds.
Transition metals are often used as catalysts in chemical reactions. Examples: the Haber Process, the decomposition of hydrogen peroxide, hydrogenation of alkenes, and hydrogenation of oils. They can be used in catalytic converters in cars and are biological catalysts. An example is heme, which is the iron center of hemoglobin. Hemoglobin is the protein that transports oxygen in the blood. Blood is red due to the presence of heme. Each subunit of hemoglobin has an iron atom at its center to which oxygen binds.
Magnetic properties of transition metals and their complexes depend on oxidation state of the metal, coordination number, and the geometry of the complex. Ferromagnetic metals are iron, cobalt and nickel. Paramagnetic materials contain unpaired electrons that behave as tiny magnets and are attracted by an external magnetic field. Diamagnetic materials do not contain unpaired electrons and therefore are repelled by external magnetic fields. See the below video.
Mission 2: Colored Complexes
Mission Objectives. You should be able to...
1. Explain the effect of the identity of the metal ion, the oxidation state of the metal, and the identity of the ligand on the color of transition metal ion complexes.
2. Explain the effect of different ligands on the splitting of the d-orbitals in transition metal complexes and colour observed using the spectrochemical series.
Before we proceed, let's figure out how to determine the oxidation state of the metal ions in a complex. Mike S. Jones to the rescue.
Mission Objectives. You should be able to...
1. Explain the effect of the identity of the metal ion, the oxidation state of the metal, and the identity of the ligand on the color of transition metal ion complexes.
2. Explain the effect of different ligands on the splitting of the d-orbitals in transition metal complexes and colour observed using the spectrochemical series.
Before we proceed, let's figure out how to determine the oxidation state of the metal ions in a complex. Mike S. Jones to the rescue.
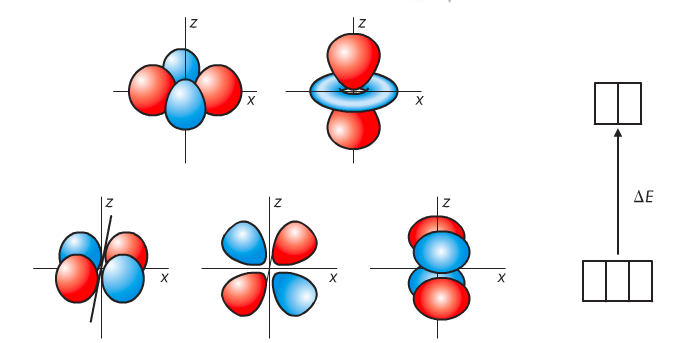
In a complex ion, d orbitals split into two sublevels. The electronic transitions between these sublevels leads to absorption and emission of photons of light, which are responsible for the color of the complex. dxy, dxz, and dyz orbitals are stable because their orbital lobes lie at 45 degrees to the Cartesian axes. dx2-y2 and dz2 orbitals are destabilized because their lobes are directed along the Cartesian axes. This is called a 3:2 split, with the last two orbitals having more energy than the first three.
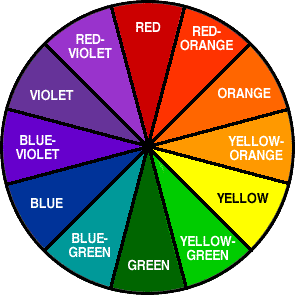
Light passes through the complex. Some of it is absorbed. The absorbed energy causes electrons to get excited and jump to the higher d orbitals. The change in energy is referred to as delta E. The color that you see is what is left behind, and is the opposite on the color wheel to what was absorbed. For instance, if you're seeing red, the absorbed energy falls in the green range on the color wheel.
The energy separation between the two split degenerate sets of orbitals is defined as the crystal field splitting energy. There are five theories, but your book focuses on Crystal Field Theory (CFT). Factors that affect CFT: (1) identity of the metal ion, (2) oxidation state of the metal ion, (3) nature of the ligands, and (4) geometry of the complex ion. You need to know the first three.
Identity of the metal ion. This influences the extent of the crystal field splitting. In general, the splitting field energy parameter increases as you go down a group with the metal in the same oxidation state. Oxidation state increases as the parameter increases. Distances between the metal and the ligands decrease, resulting in a better overlap between the metal orbitals and the ligand orbitals.
Nature of the ligands. They may have different charge densities. The greater the charge density, the greater the crystal field splitting. Ammonia's charge density is greater than that of water. Ligands can be arranged into a spectrochemical series, based on the order of increasing field energy parameters. Using the series, the further to the right, the bigger the split. A big split = a big jump = big amount of energy absorbed (and falls in the violet range). A small split = a small jump = a small amount of energy absorbed (and falls into the red range).
From the Pearson text:
Light passes through the complex. Some of it is absorbed. The absorbed energy causes electrons to get excited and jump to the higher d orbitals. The change in energy is referred to as delta E. The color that you see is what is left behind, and is the opposite on the color wheel to what was absorbed. For instance, if you're seeing red, the absorbed energy falls in the green range on the color wheel.
The energy separation between the two split degenerate sets of orbitals is defined as the crystal field splitting energy. There are five theories, but your book focuses on Crystal Field Theory (CFT). Factors that affect CFT: (1) identity of the metal ion, (2) oxidation state of the metal ion, (3) nature of the ligands, and (4) geometry of the complex ion. You need to know the first three.
Identity of the metal ion. This influences the extent of the crystal field splitting. In general, the splitting field energy parameter increases as you go down a group with the metal in the same oxidation state. Oxidation state increases as the parameter increases. Distances between the metal and the ligands decrease, resulting in a better overlap between the metal orbitals and the ligand orbitals.
Nature of the ligands. They may have different charge densities. The greater the charge density, the greater the crystal field splitting. Ammonia's charge density is greater than that of water. Ligands can be arranged into a spectrochemical series, based on the order of increasing field energy parameters. Using the series, the further to the right, the bigger the split. A big split = a big jump = big amount of energy absorbed (and falls in the violet range). A small split = a small jump = a small amount of energy absorbed (and falls into the red range).
From the Pearson text:
Oxidation state of the metal ion. The lower the oxidation state, the smaller the split because there is less interaction with the ligands.
Learn more about colored complexes with the below videos.
Learn more about colored complexes with the below videos.
Practice worksheet, courtesy of MSJChem. We will also work the problems in the text starting on page 325. Make sure you have your data booklets.




 RSS Feed
RSS Feed