Mission 1: Those Doggone Electrons
Mission Objectives. You should be able to...
1. Solve problems using E = hv
2. Calculate value of the first IE from spectral data which gives wavelength or frequency of the convergence limit.
3. Deduce the group of an element from its successive IE data.
4. Explain trends and discontinuities in data on first IE across a period.
Ionization energy is related to the process X(g) --> X+(g) + e-. Successive ionizations are possible: X+(g) --> X2+(g) + e-. This can go on, as represented by: nth = X(n-1)(g) --> Xn+ + e-.
For a given element, the ionization energy increases for successive ionizations in the order IE1 < IE2 < IE3 < IE4 < IE5... This is because it requires more and more energy to remove electrons as you get closer to the nucleus.
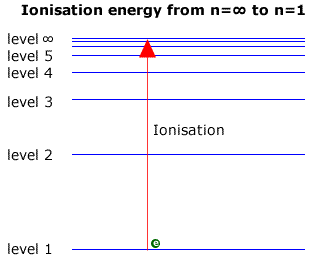
Emission lines converge at higher energies. Look at the figure below. At the limit of convergence, the lines merge and form a continuum. Beyond this limit, the electron can have any energy because it is no longer under the influence of the nucleus; the electron is outside of the atom, which means ionization has occurred.
Mission Objectives. You should be able to...
1. Solve problems using E = hv
2. Calculate value of the first IE from spectral data which gives wavelength or frequency of the convergence limit.
3. Deduce the group of an element from its successive IE data.
4. Explain trends and discontinuities in data on first IE across a period.
Ionization energy is related to the process X(g) --> X+(g) + e-. Successive ionizations are possible: X+(g) --> X2+(g) + e-. This can go on, as represented by: nth = X(n-1)(g) --> Xn+ + e-.
For a given element, the ionization energy increases for successive ionizations in the order IE1 < IE2 < IE3 < IE4 < IE5... This is because it requires more and more energy to remove electrons as you get closer to the nucleus.
Emission lines converge at higher energies. Look at the figure below. At the limit of convergence, the lines merge and form a continuum. Beyond this limit, the electron can have any energy because it is no longer under the influence of the nucleus; the electron is outside of the atom, which means ionization has occurred.
There's some problem solving that goes along with this, so you'll need your Data Booklets and calculators.
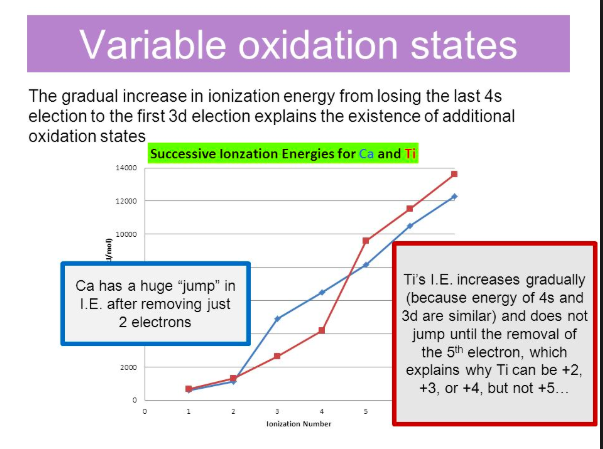
Periodic Trends in IE. The image below shows successive ionization energies for calcium and titanium. In the case of calcium, there is a significant jump going from IE2 to IE3; the third IE corresponds to the removal of an electron from the fully occupied 3p sublevel. As a result, there aren't any Ca3+ ions. Titanium exhibits oxidation states of +2, +3 and +4, with the most stable being +4. The figure shows that there is a large jump in IE for titanium going from IE4 to IE5, which corresponds to the removal of a fifth electron, supporting the observation that titanium doesn't form ions with +5 oxidation states. The IEs increase more gradually than for calcium because electrons are being removed from the 3d and 4s orbitals which are much closer in energy compared to 3p and 4s.
Image courtesy of slideplayer.
Image courtesy of slideplayer.
 RSS Feed
RSS Feed