C6: Electrochemistry & Rechargeable Batteries
In a primary electrochemical cell, the materials are consumed and the reaction is not reversible. Either the anode or the electrolyte (or both) need to be replaced, or the battery is thrown away. Typically the anode is oxidized and can no longer be used. Furthermore, the ions traveling through the cell can polarize the cell, which causes the reaction to stop. Polarization can cause a buildup of hydrogen bubbles on the anode, which increases the cell's internal resistance and reduce its output. In a secondary electrochemical cell (aka a rechargeable battery), the chemical reactions that generate electricity can be reversed by applying an electric current to them. Secondary cells can deliver stronger current demands than primary cells.
Secondary Cells: Lead-Acid Batteries
Rechargeable batteries are used in cars, for energy storage in the electric grid, in motorized electric vehicles, as emergency backup and for other uses. The typical lead-acid battery in a car is recharged while driving. In the lead-acid battery, the electrolyte is sulfuric acid.
Secondary Cells: Lead-Acid Batteries
Rechargeable batteries are used in cars, for energy storage in the electric grid, in motorized electric vehicles, as emergency backup and for other uses. The typical lead-acid battery in a car is recharged while driving. In the lead-acid battery, the electrolyte is sulfuric acid.
Secondary Cells: Nickel-Cadmium Batteries
The NiCd rechargeable cell (used in toys and electronics) has a nickel (III) oxide hydroxide cathode, which reduces to nickel (II) hydroxide during discharge and a cadmium metal anode, which is oxidized to cadmium hydroxide. The electrolyte is aqueous potassium hydroxide.
The NiCd rechargeable cell (used in toys and electronics) has a nickel (III) oxide hydroxide cathode, which reduces to nickel (II) hydroxide during discharge and a cadmium metal anode, which is oxidized to cadmium hydroxide. The electrolyte is aqueous potassium hydroxide.
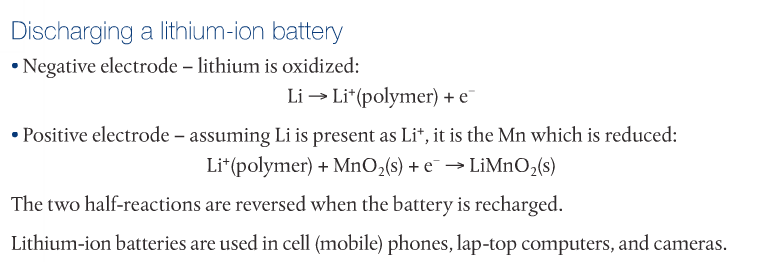
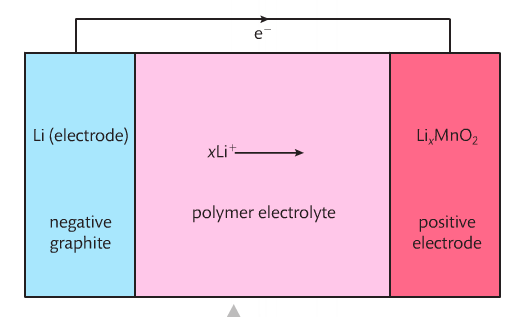
Secondary Cells: Lithium-Ion Batteries
Lithium-Ion batteries used lithium atoms absorbed into a lattice of graphite electrodes rather than lithium metal for the anode. The cathode is a lithium cobalt oxide complex. The lithium atoms are oxidized to lithium ions during discharge. Lithium is an ideal material for lightweight batteries. Lithium-ion batteries are used in cell phones, laptops and cameras.
Lithium-Ion batteries used lithium atoms absorbed into a lattice of graphite electrodes rather than lithium metal for the anode. The cathode is a lithium cobalt oxide complex. The lithium atoms are oxidized to lithium ions during discharge. Lithium is an ideal material for lightweight batteries. Lithium-ion batteries are used in cell phones, laptops and cameras.
Pages 690-691 contain tables that compare and contrast the different types of batteries.
The voltage of a battery depends on the nature of the cathode and anode. The further apart the electrode potentials of the oxidizing and reducing materials, the more voltage per cell is available. Putting cells in series provides increased voltage.
The total number of electrons moving along with the energy given to them by the cell give a measure of how much work can be done by the current. This in turn depends on the nature and quantity of the materials (mass and surface area) as well as the specific energy density.
The moving electrons in the external circuit that provide us with useful energy, but each electrochemical cell also has to move cations and anions inside the cell. A battery's internal resistance depends on ion mobility, electrolyte conductivity, and the electrode's surface area.
Reactions occur faster at higher temperatures. At lower temperatures, reactions slow down. Ion mobility is reduced and the battery's internal resistance is increased. While batteries have lower resistance at higher temperatures, they also have an increased rate of self-discharge, so storing batteries at high temperatures is not a good idea.
The voltage of a battery depends on the nature of the cathode and anode. The further apart the electrode potentials of the oxidizing and reducing materials, the more voltage per cell is available. Putting cells in series provides increased voltage.
The total number of electrons moving along with the energy given to them by the cell give a measure of how much work can be done by the current. This in turn depends on the nature and quantity of the materials (mass and surface area) as well as the specific energy density.
The moving electrons in the external circuit that provide us with useful energy, but each electrochemical cell also has to move cations and anions inside the cell. A battery's internal resistance depends on ion mobility, electrolyte conductivity, and the electrode's surface area.
Reactions occur faster at higher temperatures. At lower temperatures, reactions slow down. Ion mobility is reduced and the battery's internal resistance is increased. While batteries have lower resistance at higher temperatures, they also have an increased rate of self-discharge, so storing batteries at high temperatures is not a good idea.





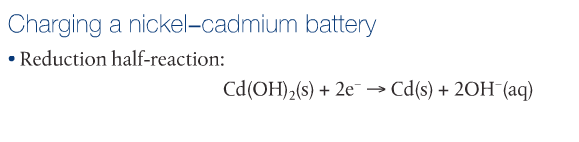

 RSS Feed
RSS Feed