C6 Part 2: Fuel Cells
A fuel cell is an electrochemical device that converts chemical potential energy in a fuel into electrical energy. In a hydrogen fuel cell, the fuel is hydrogen (obviously), which is oxidized by oxygen and produces water. There is therefore no pollution and fuel cells are very efficient. The key components of a fuel cell are (1) the electrolyte or separator which prevents components from mixing. The proton exchange membrane is a polymer which allows H+ ions to diffuse but not electrons or molecules, (2) the oxidizing and reducing electrodes which are catalysts that allow the chemical reaction to occur, and (3) the bipolar plate which collects the current and builds up voltage in the cell.
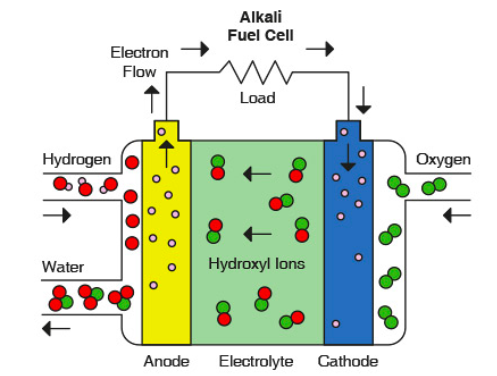
Alkali Fuel Cells. The electrolyte in these cells is a solution of potassium hydroxide, providing a source of OH- ions. As the OH- ions migrated towards the anode, they reacted with H+ ions, producing water. If an acidic electrolyte (such as phosphoric acid) is used, then the H+ ions will migrate towards the cathode.
Image courtesy of alkammonia.eu.
Alkali Fuel Cells. The electrolyte in these cells is a solution of potassium hydroxide, providing a source of OH- ions. As the OH- ions migrated towards the anode, they reacted with H+ ions, producing water. If an acidic electrolyte (such as phosphoric acid) is used, then the H+ ions will migrate towards the cathode.
Image courtesy of alkammonia.eu.
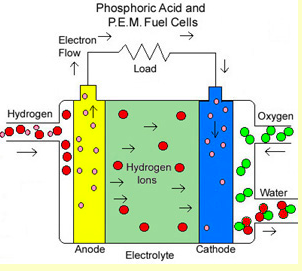
Hydrogen fuel cells use hydrogen and oxygen as fuel. The heat formed can be used as a heat source. However, the hydrogen has to be pure and often platinum or other catalysts are added to graphite electrodes, which makes them expensive to run on a commercial scale. The oxygen comes from the air. The hydrogen comes from (1) the electrolysis of water, or (2) reforming hydrocarbons or biofuels.
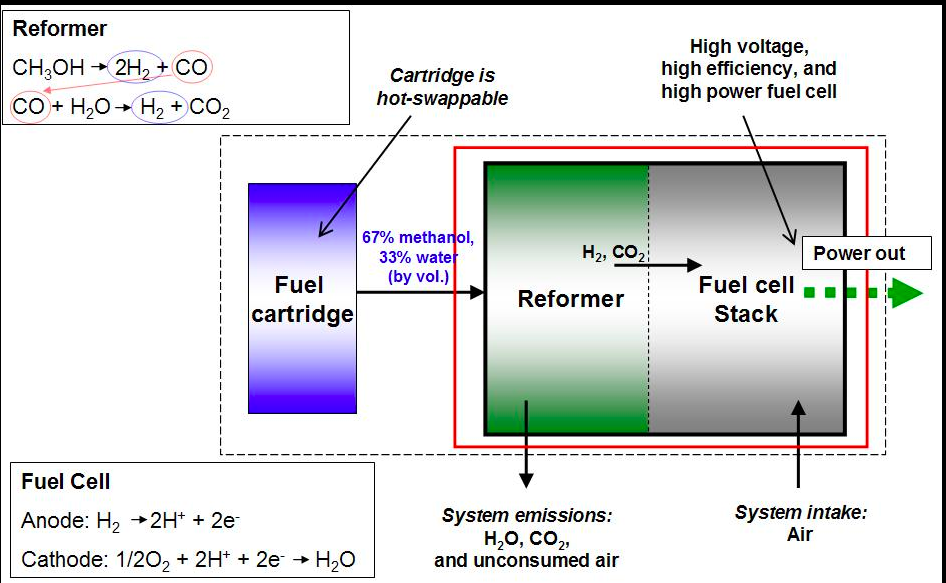
Direct Methanol Fuel Cells. Methanol is used as a fuel instead of hydrogen. Methanol provides H+ ions at the anode. The fuel cell has the same components as the hydrogen PEM fuel cell. The anode reaction requires water, so a dilute solution of 1M methanol is used.
Images courtesy of wikipedia.org and americanhistory.si.edu
Direct Methanol Fuel Cells. Methanol is used as a fuel instead of hydrogen. Methanol provides H+ ions at the anode. The fuel cell has the same components as the hydrogen PEM fuel cell. The anode reaction requires water, so a dilute solution of 1M methanol is used.
Images courtesy of wikipedia.org and americanhistory.si.edu
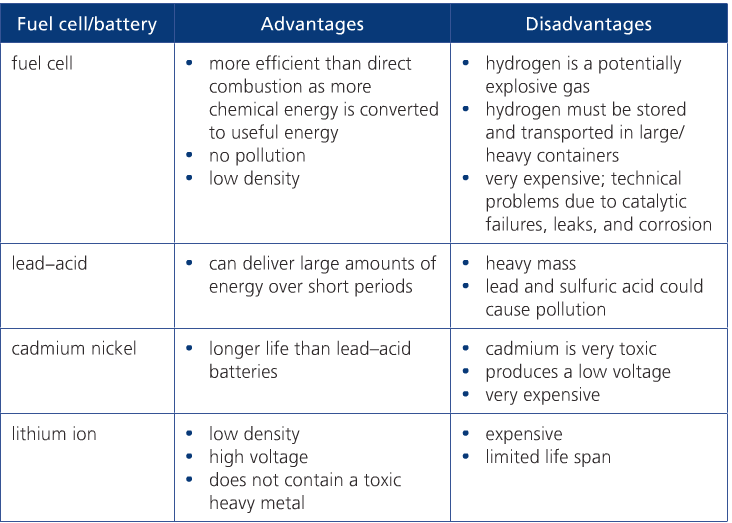
Page 695 in your text compares different fuel cells based on their energy density and specific energy. The table below, from the Pearson textbook, compares the advantages and disadvantages of different batteries and fuel cells.
While octane has a high energy density, fuel cells tend to have a higher thermodynamic efficiency. This is the ratio of the Gibbs energy change to the enthalpy change. (delta G/delta H).
It is possible to alter the EMF of a cell by changing the concentration of the mobile ions. When standard conditions exist, it is possible to predict the EMF of a voltaic cell by adding their half-cell potentials under standard conditions.
The Nernst equation can be used to calculate the potential of an electrochemical cell under non-standard conditions.
Mr. Thornley is on the case.
It is possible to alter the EMF of a cell by changing the concentration of the mobile ions. When standard conditions exist, it is possible to predict the EMF of a voltaic cell by adding their half-cell potentials under standard conditions.
The Nernst equation can be used to calculate the potential of an electrochemical cell under non-standard conditions.
Mr. Thornley is on the case.
 RSS Feed
RSS Feed