Types of Organic Reactions
Mission 1: Types of Organic Reactions
Mission Objectives. You should be able to...
1. Explain why OH- is a better nucleophile than water.
2. Deduce the mechanism of the nucleophilic substitution reactions with aqueous NaOH in terms of SN1 and SN2 mechanisms.
3. Explain how the rate depends on the identity of the halogen (leaving group) whether the halogenoalkane is primary, secondary or tertiary.
4. Outline the difference between protic and aprotic solvents.
Nucleophiles are species that are electron-rich, which means that they have a lone pair of electrons and may carry a negative charge. They are attracted to carbon and act as Lewis bases and can donate electrons. Examples include: OH-, H2O, NH3, and CN-.
Electrophiles are reactants that are electron-deficient, as they have a positive charge or partial positive charge. They are attracted to benzene and act as Lewis acids. Examples include: H+, Br+, and NO2+.
When dealing with these kinds of reactions, we must determine (1) what happens, which is the type of reaction, and (2) how it happens, which is the mechanism of the reaction.
Mission Objectives. You should be able to...
1. Explain why OH- is a better nucleophile than water.
2. Deduce the mechanism of the nucleophilic substitution reactions with aqueous NaOH in terms of SN1 and SN2 mechanisms.
3. Explain how the rate depends on the identity of the halogen (leaving group) whether the halogenoalkane is primary, secondary or tertiary.
4. Outline the difference between protic and aprotic solvents.
Nucleophiles are species that are electron-rich, which means that they have a lone pair of electrons and may carry a negative charge. They are attracted to carbon and act as Lewis bases and can donate electrons. Examples include: OH-, H2O, NH3, and CN-.
Electrophiles are reactants that are electron-deficient, as they have a positive charge or partial positive charge. They are attracted to benzene and act as Lewis acids. Examples include: H+, Br+, and NO2+.
When dealing with these kinds of reactions, we must determine (1) what happens, which is the type of reaction, and (2) how it happens, which is the mechanism of the reaction.
The partial positive charge makes carbon atoms electron-deficient and therefore susceptible to attack by nucleophiles, which are electron-rich species capable of donating a pair of electrons to form covalent bonds. There are two types of nucleophilic substitution: SN1 and SN2. During these reactions, the carbon-halogen bond breaks and the halogen atom is released as a negative ion. This type of bond breakage is known as heterolytic fission.
The halogen, because it detaches, is known as the leaving group. The mechanism depends on whether the halogenoalkane is primary, secondary or tertiary.
Recall that primary compounds are attached to the functional group and at least two hydrogens. Secondary molecules are attached to the functional group, one hydrogen and two alkyl groups. Tertiary molecules are attached to the functional group and three alkyl groups, but no hydrogen atoms.
SN2 Reactions and primary halogenoalkanes. Nucleophilic substitution in primary halogenoalkanes proceed in one step. The slow step involves both the halogenoalkane and the nucleophile so that the rate of reaction is dependent on the concentrations of both reactants: rate = k[halogenoalkane][nucleophile] The molecularity is bimolecular, as there are two molecular entities.
When drawing mechanisms for SN2 reactions, please follow the rules on page 440. They are serious about those curly arrows. Below is a video showing the mechanism for a SN2 reaction using bromomethane in a solution with hydroxide ions.
The halogen, because it detaches, is known as the leaving group. The mechanism depends on whether the halogenoalkane is primary, secondary or tertiary.
Recall that primary compounds are attached to the functional group and at least two hydrogens. Secondary molecules are attached to the functional group, one hydrogen and two alkyl groups. Tertiary molecules are attached to the functional group and three alkyl groups, but no hydrogen atoms.
SN2 Reactions and primary halogenoalkanes. Nucleophilic substitution in primary halogenoalkanes proceed in one step. The slow step involves both the halogenoalkane and the nucleophile so that the rate of reaction is dependent on the concentrations of both reactants: rate = k[halogenoalkane][nucleophile] The molecularity is bimolecular, as there are two molecular entities.
When drawing mechanisms for SN2 reactions, please follow the rules on page 440. They are serious about those curly arrows. Below is a video showing the mechanism for a SN2 reaction using bromomethane in a solution with hydroxide ions.
Quick summary of SN2 mechanism: Bimolecular nucleophilic substitution. Involves heterolytic fission and nucleophilic substitution with primary halogenoalkanes. An unstable state is created, which makes the reaction bimolecular. Both incoming and leaving groups happen simultaneously (one step) and both are responsible for the rate-determining step.
SN1 Reactions and tertiary halogenoalkanes. Tertiary halogenoalkanes undergo nucleophilic substitution reactions that involve more than one step. The rate-determining step involves only the halogenoalkane; the bond to the leaving group breaks, forming a carbocation. A carbocation is a carbon atom with a slightly positive charge. The reaction is first order: rate = k[halogenoalkane]. The reaction is unimolecular.
When drawing mechanisms for SN1 reactions, please follow the rules on page 441.
Below is a video that goes through the SN1 mechanism using terbutylbromide and methanol as a solvent.
SN1 Reactions and tertiary halogenoalkanes. Tertiary halogenoalkanes undergo nucleophilic substitution reactions that involve more than one step. The rate-determining step involves only the halogenoalkane; the bond to the leaving group breaks, forming a carbocation. A carbocation is a carbon atom with a slightly positive charge. The reaction is first order: rate = k[halogenoalkane]. The reaction is unimolecular.
When drawing mechanisms for SN1 reactions, please follow the rules on page 441.
Below is a video that goes through the SN1 mechanism using terbutylbromide and methanol as a solvent.
Summary of steps for SN1 mechanism: (1) formation of the carbocation, (2) nucleophilic attack, and (3) deprotonation.
Below, Richard Thornley briefly explains rates of nucleophilic substitution using the halogens. The second video reviews the steps in SN1 and SN2 reactions.
Below, Richard Thornley briefly explains rates of nucleophilic substitution using the halogens. The second video reviews the steps in SN1 and SN2 reactions.
Factors that affect the rate of nucleophilic substitution:
1. The identity of the halogen. The quicker the rate-determining step, the higher the rate of reaction and a better leaving group achieves this. As you move down G17, the strength of the carbon-halogen bond decreases as the size of the halogen increases. A larger halogen results in long, weak bonds. Therefore iodine is the better leaving group out of all the halogens.
2. The classes of halogenoalkane. The class of halogenoalkane has a direct effect on the rate of formation. Tertiary carbocations have greater stability than primary carbocations. They rapidly form and reacts with the nucleophile immediately.
3. The choice of solvent. SN2 reactions are best performed in aprotic, polar solvents and SN1 reactions are best performed in protic, polar solvents. Aprotic polar solvents are ideal for SN2 reactions because they don't possess O-H or N-H groups and cannot solvate the nucleophile. Aprotic solvents include ethyl ethanoate and propanone. Protic polar solves are suitable for SN1 reactions because they are polar in nature, possess either an O-H or N-H groups so H bonds can be formed, and solvate the nucleophile, thus inhibiting its ability to attack electrophiles.
1. The identity of the halogen. The quicker the rate-determining step, the higher the rate of reaction and a better leaving group achieves this. As you move down G17, the strength of the carbon-halogen bond decreases as the size of the halogen increases. A larger halogen results in long, weak bonds. Therefore iodine is the better leaving group out of all the halogens.
2. The classes of halogenoalkane. The class of halogenoalkane has a direct effect on the rate of formation. Tertiary carbocations have greater stability than primary carbocations. They rapidly form and reacts with the nucleophile immediately.
3. The choice of solvent. SN2 reactions are best performed in aprotic, polar solvents and SN1 reactions are best performed in protic, polar solvents. Aprotic polar solvents are ideal for SN2 reactions because they don't possess O-H or N-H groups and cannot solvate the nucleophile. Aprotic solvents include ethyl ethanoate and propanone. Protic polar solves are suitable for SN1 reactions because they are polar in nature, possess either an O-H or N-H groups so H bonds can be formed, and solvate the nucleophile, thus inhibiting its ability to attack electrophiles.
What are protic and aprotic solvents? Let's find out.
Read up on electrophilic reactions (p. 445-447). Below is a short video on Markovnikov's rule.
Synthetic Routes
Mission 2: Synthetic Routes
Mission Objectives. You should be able to...
1. Deduce a multi-step synthetic route given starting reagents and the products.
Mission Objectives. You should be able to...
1. Deduce a multi-step synthetic route given starting reagents and the products.
Mission 3: Stereoisomers
Mission Objectives. You should be able to...
1. Construct 3D models of a wide range of stereoisomers
2. Explain stereoisomerism in non-cyclic alkenes and C3 C4 cycloalkanes.
3. Compare physical and chemical properties of enantiomers.
4. Describe and explain optical isomers in simple organic molecules.
5. Distinguish between optical isomers using a polarimeter.
Mission Objectives. You should be able to...
1. Construct 3D models of a wide range of stereoisomers
2. Explain stereoisomerism in non-cyclic alkenes and C3 C4 cycloalkanes.
3. Compare physical and chemical properties of enantiomers.
4. Describe and explain optical isomers in simple organic molecules.
5. Distinguish between optical isomers using a polarimeter.
Stereoisomers
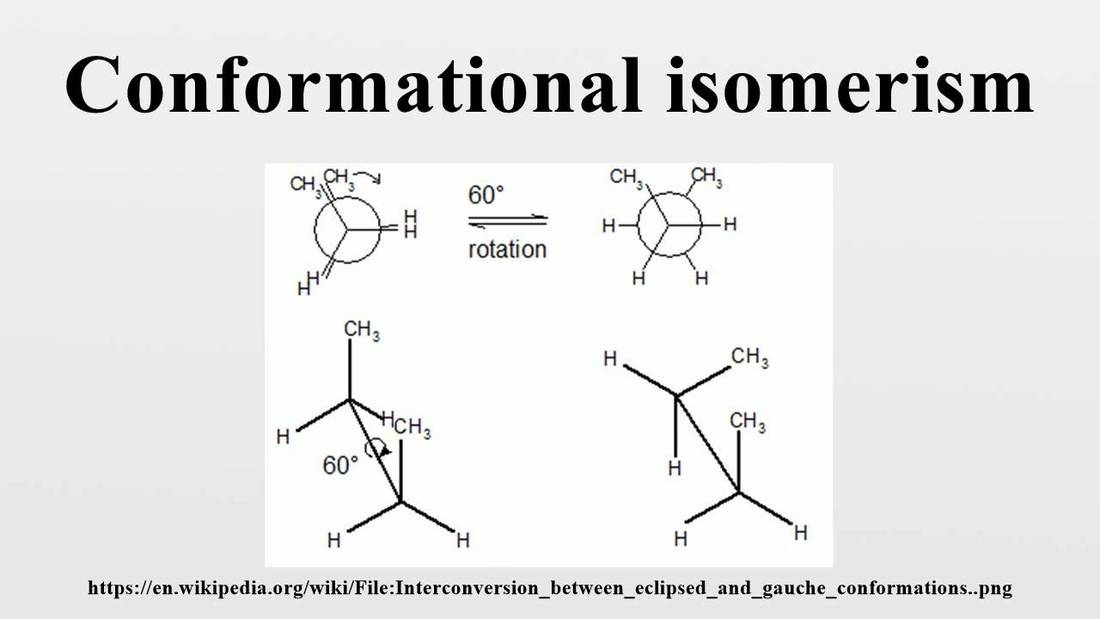
Stereoisomers have an identical molecular formula and bond multiplicity but show different spatial arrangements of the atoms. Stereoisomers can be subdivided into two major classes: conformational isomers and configurational isomers. Conformational isomers can be interconverted by rotation about the sigma bond without breaking any bonds. But if there is a double bond present, the pi bond must be broken. Therefore conformational isomers differ from one another in the arrangement of atoms around a single bond. We will play with the ethane molecule and reference page 453.
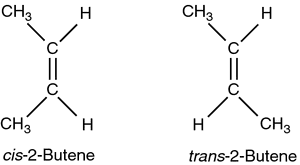
| Configurational isomers can only be interconverted by the breaking of the sigma or pi bond or through rearrangement of the stereocenters (an atom with 3 or more different attachments). Configurational isomers are subdivided into cis-trans and E/Z isomers on one end and optical isomers on the other. In terms of nomenclature, a special set of rules are required. Read about it on page 454. Cis-isomers are the equivalent of Z isomers and trans-isomers are the equivalent of E isomers. | Cis-trans isomers are determined by the positions of substituents (terminal atoms or groups of atoms in a terminal position) relative to a reference plane. For alkenes, the reference plane is the C=C. Cis-isomers have substituents one the SAME SIDE of the reference plane whereas trans-isomers have substituents on OPPOSITE SIDES of the reference plane. |
Optical Isomerism is a type of configurational isomerism determined by the presence of chiral carbon atoms, also known as a stereocenter or asymmetric center. A chiral carbon is bonded to four different atoms or groups of atoms. Optical isomers have the ability to rotate plane-polarized light and exist in pairs that are called enantiomers or diastereomers. Enantiomers are non-superimposable mirror images of each other. They have no plane or symmetry and their optical activity is readily assigned when the molecules are represented as 3-D images.
The physical and chemical properties of two enantiomers of a particular substance are the same (BP, MP, viscosity, density, and solubility). Many chemical properties are the same except for their chemical reactions with other optical isomers. Enzymes in the body are chiral. Read about the examples of + and - limonene and the drug thalidomide on page 457.
Diastereomers are non-superimposable but they do not form mirror images. They have two or more stereocenters and differ in the configuration of at least one center. Diastereomers with the same general formula have different physical and chemical properties.
The physical and chemical properties of two enantiomers of a particular substance are the same (BP, MP, viscosity, density, and solubility). Many chemical properties are the same except for their chemical reactions with other optical isomers. Enzymes in the body are chiral. Read about the examples of + and - limonene and the drug thalidomide on page 457.
Diastereomers are non-superimposable but they do not form mirror images. They have two or more stereocenters and differ in the configuration of at least one center. Diastereomers with the same general formula have different physical and chemical properties.
 RSS Feed
RSS Feed