Click HERE for Missions.
|
Housekeeping: We are now studying physics. The units we will cover are kinematics and wave basics. You have a project at the end of the unit where you have to construct a race track and measure the velocity and acceleration of a Hot Wheels car.
Click HERE for Missions.
0 Comments
Housekeeping: This week and the week after Spring Break, you will work on this quarter's Criterion D project. Details are below.
You will take your test over Energy on Thursday of this week. It has two parts: 20 MC on Socrative and problem solving by hand. We will review in class tomorrow, but make sure you start studying. Click HERE for Energy Research Project. Housekeeping: Welcome Back and Merry New Year!!! We will begin 2019 with a unit on stoichiometry. Click HERE for Missions.
Chemical Reactions that Changed the World
You and a partner will investigate one of the following chemical reactions. You must be able to explain what the reaction is (not necessarily an equation), what it produces, and how it changed the world. Speculate on what our world would be like without these reactions. You will do this as a PowerPoint presentation (10 slides max). The presentation is due December 19, 2018, but you will email me the slide show the night before. I will give you a presentation rubric that you must follow. This will be graded under Criterion A & D. Topics: Yuki & Kevin Saponification Thariq & Nik Polymerization of polyethylene Na Young & Efa Haber Process Ciara & Sasha Maillard Reaction James & Max Combustion of crude oil Brian & Gilbert Fat Hydration Shaneta & Yujin Creation of Gunpowder Ica & Yi-Jen Fermentation Sabrina & Felicia Penicillin Jeanice & Vianna Pasteurization Housekeeping: We are now investigating chemical reactions. You will have a quiz overt chemical bonding Nov. 14 and your exam over chemical bonding is scheduled for Nov. 28. Included in this new unit are two labs where you will investigate different kinds of chemical reactions. One you will design yourself. I'll explain more about this next week.
Click HERE for Missions Design Lab: Over the course of next week, you will design a lab to investigate single replacement and double replacement reactions. Recall the structure of a single-replacement chemical equation. You will perform these experiments in test tubes and predict then observe the outcomes. You will document evidence that a reaction takes place. Next week, you will finish up SRRs. This will be graded under Criterion B and C. You will submit the following: 1. Completed data table, a paragraph describing what you learned, and a detailed evaluation that includes strengths, weaknesses and ways to improve the lab. Each of these are worth up to 8 points, depending on the quality. (Criterion C) THIS IS DUE TODAY (November 28th). 2. A writeup that includes (1) research question, (2) a tentative hypothesis (relate this to the activity series of metals), (3) IV/DV/Control, (4) materials list (bulleted), (5) numbered step-by-step procedure, and (6) how the data should be collected. (Criterion B) THIS IS DUE TOMORROW (November 29th). Because you worked in groups, the smartest thing to do is to divvy up the workload. Before you submit (via dropbox), show it to me so that I can give you feedback. Housekeeping: Your test over periodicity is scheduled for October 24.
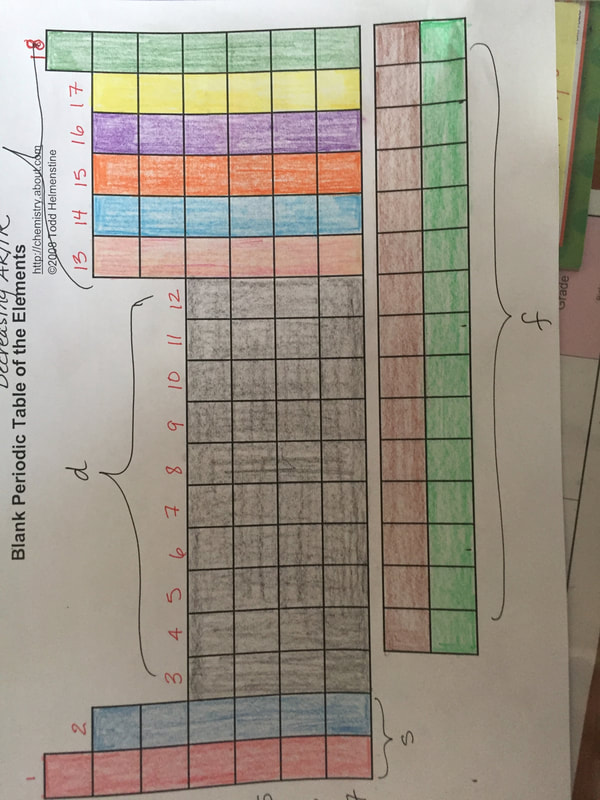
We are now studying chemical bonding. This unit will take anywhere between 3 to 5 weeks to cover, depending on how well you learn the objectives. Before we start talking about ion formation, we need to discuss the behavior of transition metals. We will focus on period 4 d-block elements: scandium through zinc. Click HERE for Missions. Today you will show your understanding of periodic table structure. You will be given a blank periodic table and some color pencils. You must do the following:
1. Color the alkali metals red 2. Color the alkaline earth metals blue 3. Color the noble gases green 4. Color the halogens yellow 5. Color the chalcogens purple 6. Color the prictogens orange 7. Color the transition metals black 8. Color the carbonicles light blue 9. Color the boronites pink 10. Color the lanthanides brown 11. Color the actinides light green. 12. Using arrows, identify the 4 periodic trends you studied: ionization energy, atomic radius, ionic radius and electronegativity 13. Identify the 4 blocks 14. Number the groups 15. Number the periods Each item is worth one point. You have until the end of class to complete this activity. Drop it in my inbox before you leave. Answers below Housekeeping: We are now technically finished with the atom. Today (9/25/18), we will do some simple activities to supplement what you've learned so far before moving into periodicity. Your test over atomic structure is scheduled for October 3rd.
Your cereal box is due tomorrow. We are now investigating periodicity. This is a short chapter that goes through the full structure of the periodic table and the names of all the elemental families. Some of the content should be familiar to you. We will be done with periodicity before fall break, which means your test will be after the break. Click HERE for Missions. Housekeeping: This week, you need to take a quiz. Thursday is your short day, so that's when we will do it. We also need to schedule the exam date.
We have completed the Matter unit and will move into atomic structure this week. Everything is going well so far. Any questions? Week of 9/17-21: You will create a cereal box over an assigned element. Grading rubric is here. You will be given one double period to work on the cereal box and everything else must be completed outside of class. The cereal box is due September 26. CLICK HERE for Missions. |


 RSS Feed
RSS Feed