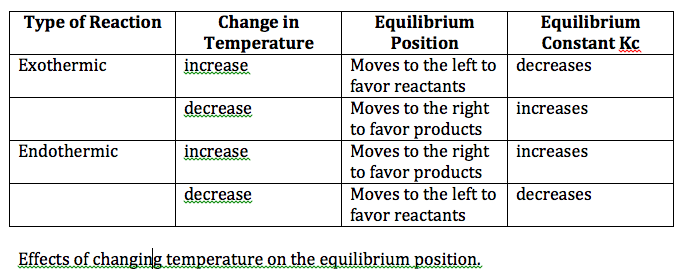
Chapter 17: Equilibrium
Understandings:
1. Le Chatelier's Principle for changes in concentration can be explained by the equilibrium law.
2. The position of equilibrium corresponds to a maximum value of entropy and a minimum in the value of Gibbs' Free Energy.
3. The Gibbs Free Energy change of a reaction and the equilibrium constant can both be used to measure the position of an equilibrium reaction and are related by the equation: delta G = -R ln K
Mission 1: Equilibrium Law
Mission Objectives. You should be able to...
1. Solve homogeneous equilibrium problems using the expression for Kc.
2. Relationship between delta G and the equilibrium constant.
3. Perform calculations using delta G = -RT ln K
The below video from Richard Thornley demonstrates equilibrium in a qualitative fashion. Watch it.
1. Le Chatelier's Principle for changes in concentration can be explained by the equilibrium law.
2. The position of equilibrium corresponds to a maximum value of entropy and a minimum in the value of Gibbs' Free Energy.
3. The Gibbs Free Energy change of a reaction and the equilibrium constant can both be used to measure the position of an equilibrium reaction and are related by the equation: delta G = -R ln K
Mission 1: Equilibrium Law
Mission Objectives. You should be able to...
1. Solve homogeneous equilibrium problems using the expression for Kc.
2. Relationship between delta G and the equilibrium constant.
3. Perform calculations using delta G = -RT ln K
The below video from Richard Thornley demonstrates equilibrium in a qualitative fashion. Watch it.
In order to solve equilibrium problems, you must be able to complete an ICE Chart. ICE stands for "Initial, Change, Equilibrium." Below you will find two videos. The first one shows you how to construct an ICE Chart and the second video shows you how to do the algebra required to solve for equilibrium. You need to take notes on this.
In your textbook, you have worked examples in the purple boxes. Use them and the below video as a guide. After the video, you will see some practice problems that you must complete.
Gibbs' Free Energy (G) describes the spontaneity and temperature dependence of a reaction. The free energy will change as reactants are converted into products. The reaction will be spontaneous in the direction that results in a decrease in free energy.
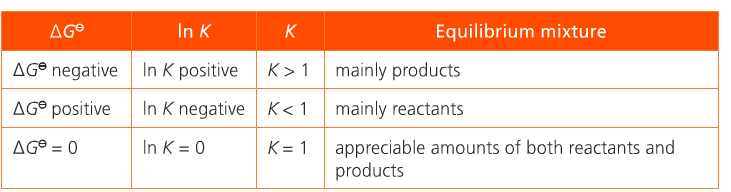
When K is determined for a given reaction, its value indicates whether products or reactants are favored at equilibrium. Delta G for a given reaction is an indication of whether the forward or reverse reaction is favored. See below. Commit to memory.
When K is determined for a given reaction, its value indicates whether products or reactants are favored at equilibrium. Delta G for a given reaction is an indication of whether the forward or reverse reaction is favored. See below. Commit to memory.
At a given temperature, a negative delta G value for a reaction indicates that the reaction is spontaneous and the equilibrium concentrations of the products are larger than the equilibrium concentrations of the reactant and K > 1. The more negative the value of delta G, the more the forward reaction is favored and the larger the value of K.
The relationship between standard Gibbs' free energy change, temperature, and K is as follows:
delta G = -RT ln K
where R is the gas constant (8.31 J/K mol), T is temperature in Kelvins, and ln K is the natural log of the equilibrium constant. This equation is in your Data Booklet. See the worked example on page 393.
The relationship between standard Gibbs' free energy change, temperature, and K is as follows:
delta G = -RT ln K
where R is the gas constant (8.31 J/K mol), T is temperature in Kelvins, and ln K is the natural log of the equilibrium constant. This equation is in your Data Booklet. See the worked example on page 393.
From the Pearson text: