Potential energy is stored energy.
 Source: Wikimedia Source: Wikimedia
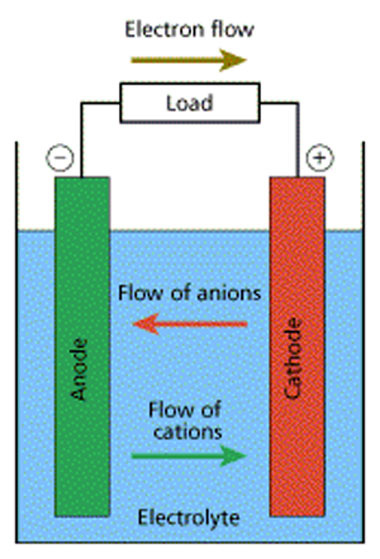
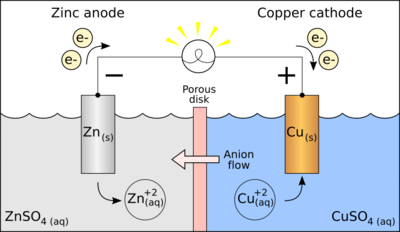
Electrochemistry transfers the chemical energy of a redox reaction into electrical energy. Recall what happens in redox reactions. When the substances involved in oxidation and reduction half–reactions are physically separated, it is called an electrochemical cell. Each half reaction occurs on the surface of an electrically conductive solid called an electrode, and each electrode is immersed in a solution containing the ions needed for the half–reaction. The electrodes are connected by a wire so that electrons can move from the oxidation half–reaction to the reduction half–reaction. The solutions are connected by a salt bridge so that ions can move between solutions. In an electrochemical cell the chemical potential energy can be harnessed as the substances undergoing oxidation push electrons through the wire to the substances undergoing reduction. Electrochemical Cells: A characteristic of electrochemical cells is that the redox reaction may occur spontaneously (voltaic cell), or non-spontaneous reactions can be forced to occur (electrolytic cell & electroplating). Electroplating is a process that coats a material with a layer of metal based on metallic activity. Voltaic cells are typically used to produce electrical energy. Batteries are a voltaic cell. Electrolytic cells use electricity to bring about a redox reaction that would normally be non-spontaneous. In other words, low-energy reactants become high-energy products. Rechargeable batteries: Spontaneous redox reactions eventually deplete the electrons available at the anode (e.g. causing a battery to become “dead”). The redox reactions can be reversed by a non-spontaneous reaction. More on batteries. Eventually, the potential in the voltaic cells will deplete because the electrons from the anode (oxidation) are depleted and built up at the cathode (reduction). Outside voltage can be connected to the voltaic cell so that the anode is connected to the negative terminal (supplying electrons that were lost) and the cathode is connected to the positive terminal (removing the electrons that were gained). |
In a voltaic cell, electrons are spontaneously emerging at the cathode so that reduction can occur. Electrons are the source of potential in a voltaic cell. The electrons are pushed from the anode towards the cathode, supplying the resistance. Since the electrons leave the anode, it is designated with a negative sign. The electrons build up at the cathode, therefore, the cathode is designated with a positive sign.
Anode: The anode is the electrode in the electrochemical cell where oxidation occurs. This is because it is losing electrons in the half-reaction. Cathode: The cathode is the electrode in the electrochemical cell where reduction occurs. This is because it is gaining electrons in the half reaction. Salt Bridge: The salt bridge completes the circuit by moving ions into the solutions to maintain electric neutrality without mixing them. |