7.4: Cellular Transport
|
Diffusion: According to the text, this is the net movement of particles from an area of high concentration to an area of low concentration. Think of how scents and fragrances make their way to your nose. If someone sprays perfume, the scent is strongest near the bottle, but in time, the scent particle will make their way to your nose. At some point, there will be an even distribution of scent particles in the air. This is diffusion.
Facilitated diffusion: When substances move through the plasma membrane via a water-filled transport protein (called a channel protein). Channel proteins open and close to allow necessary substances to diffuse through the membrane. Passive & Active Transport
Passive transport is basically movement of substances that does not require energy. Diffusion, osmosis, and facilitated diffusion are forms of passive transport. By contrast, active transport requires energy to move substances into and out of the cell. The proteins embedded in the plasma membrane are positioned so that half lies inside the bilayer and half outside the bilayer. They must work against a concentration gradient. This means instead of substances (usually ions) moving from areas of high concentration to areas of low concentration, the proteins must work in reverse: moving ions from low concentration to high concentration. This is especially true of neurons; they must pump K+, Na+, and Cl- in and out in order to fire (or synapse). Because they are working against the concentration gradient, they require a form of chemical energy called adenosine triphosphate (ATP) to assist in the process. |
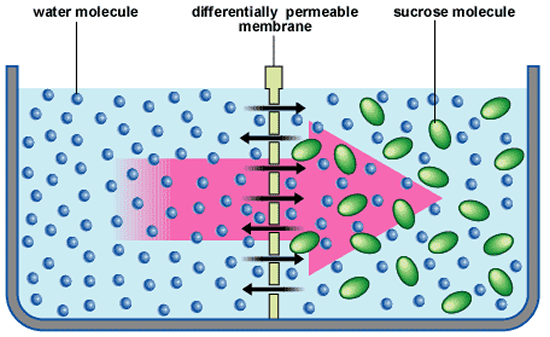
Osmosis: This is a form of specialized diffusion of water across a selectively permeable membrane. Usually the water is mixed with a solute (sometimes sugar or salt). The water can pass through the membrane but the solute cannot. The water will diffuse through the membrane towards the side with the greater concentration of solute particles until dynamic equilibrium is reached (when the concentration of the solution is the same on both sides of the membrane).
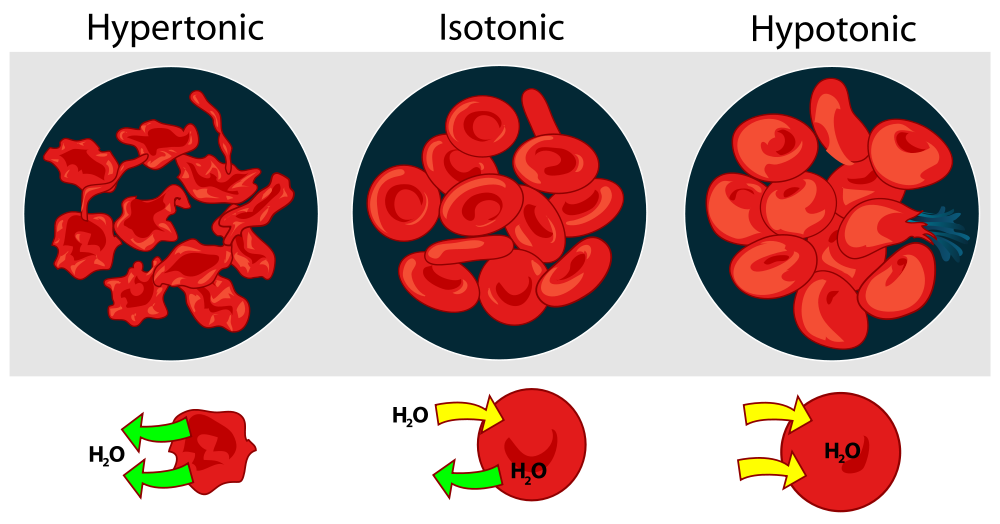
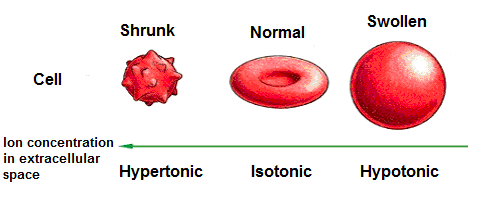
For a cell to survive, ion concentrations need to be the same on both sides of the cell membrane. If the cell does not pump out all of its extra ions to even out the concentrations, the water is going to move in. This can be very bad. The cell can swell up and explode. The classic example of this type of swelling happens when red blood cells are placed in water. The water rushes in to the cells, they expand and eventually rupture (Biology4kids.com). Tonicity
Tonicity is a measure of the osmotic pressure gradient. Cells can be immersed in tonic solutions. There are three variations: hypotonic, hypertonic, and isotonic. Hypotonic: The solution the cell is in has a lower concentration of solutes (ions, sugars, proteins, etc). There is more water outside of the cell than inside, so it flows into the cell. See the image to the left. Too much water flowing in causes the cells to burst. Hypertonic: The solution the cell is in has a higher concentration of solutes (ions, sugars, proteins, etc). There is more water inside of the cell than outside, so it flows out of the cell. See the image to the left. Too much water flowing out causes the cells to shrink. Isotonic: The solution has an equal concentration of water and solutes (ions, sugars, proteins, etc.) inside and outside of the cell, and moves in and out at an even rate. The cell retains its size. Most cells are in isotonic solutions. |
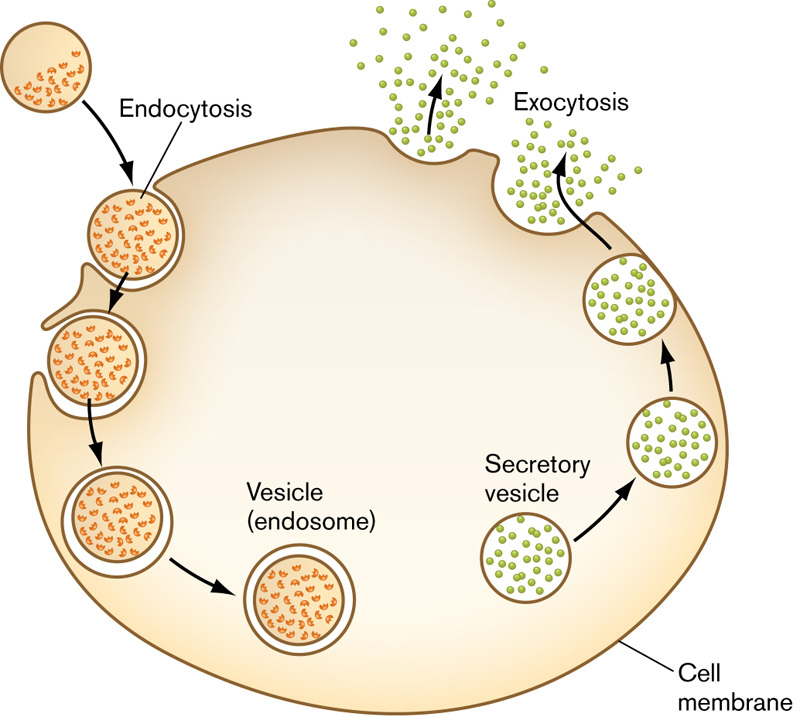
Endocytosis is the process by which a cell encloses a substance in a portion of the plasma membrane. It then pinches off inside the cell and forms a vacuole. Exocytosis is the opposite process. Materials are secreted or expelled from the cell at the plasma membrane. Both processes require energy and are used for the transport of large particles. See the below image (Courtesy of WW Norton & Company).